Crosslinking of N-acetyllactosamine-containing glycoproteins to galectin-1 with an introduced cysteine using a photoactivatable sulfhydryl reagent.
Mayumi Tamura, Takanori Igarashi, Ken-ichi Kasai, Yoichiro Arata
文献索引:Biochem. Biophys. Res. Commun. 390(3) , 581-4, (2009)
全文:HTML全文
摘要
Relatively weak interactions between galectins and their potential ligands can hinder identification of physiological lectin ligands using conventional methods such as affinity purification. We have employed a combination of cysteine mutagenesis with chemical crosslinking using a photoactivatable sulfhydryl reagent benzophenone-4-maleimide to obtain a covalent complex between human galectin-1 and the model glycoprotein ligands asialofetuin and laminin which contain an N-acetyllactosamine structure. A crosslinked product was obtained only when galectin-1 with an introduced cysteine interacted with these glycoproteins via their carbohydrate moiety. This procedure should be useful for the detection of important, and as yet unidentified, ligands for galectins which cannot be currently detected because of their relatively weak interaction.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
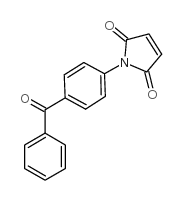 |
4-(N-马来酰亚胺基)二苯甲酮
CAS:92944-71-3 |
C17H11NO3 |
|
Subunit interactions in the clathrin-coated vesicle vacuolar...
1999-10-08 [J. Biol. Chem. 274(41) , 28909-15, (1999)] |
|
Using a low denaturant model to explore the conformational f...
2012-02-21 [Biochemistry 51(7) , 1369-79, (2012)] |
|
A cross-linking study of the N-terminal extension of human c...
2003-09-02 [Biochemistry 42(34) , 10324-32, (2003)] |
|
Formation and properties of smooth muscle myosin 20-kDa ligh...
1991-08-01 [Arch. Biochem. Biophys. 288(2) , 584-90, (1991)] |
|
Identification of the site of photocross-linking formed in t...
1991-02-05 [J. Biol. Chem. 266(4) , 2272-5, (1991)] |