Conformational effects of the non-natural alpha-methylserine on small peptides and glycopeptides.
Alberto Fernández-Tejada, Francisco Corzana, Jesús H Busto, Alberto Avenoza, Jesús M Peregrina
文献索引:J. Org. Chem. 74(24) , 9305-13, (2009)
全文:HTML全文
摘要
The synthesis and the conformational analysis in aqueous solution of a peptide and a glycopeptide containing the sequence threonine-alanine-alanine (Thr-Ala-Ala) are reported. Furthermore, the threonine residue has been replaced by the quaternary amino acid alpha-methylserine (MeSer) and their corresponding non-natural peptide and glycopeptide are also studied. The conformational analysis in aqueous solution combines NOEs and coupling constants data with Molecular Dynamics (MD) simulations with time-averaged restraints. The study reveals that the beta-O-glycosylation produces a remarkable and completely different effect on the backbone of the peptide derived from Thr and MeSer. In the former, the beta-O-glycosylation is responsible for the experimentally observed shift from extended conformations (peptide) to folded ones (glycopeptide). In contrast, the beta-O-glycosylation of the MeSer-containing peptide, which clearly shows two main conformations in aqueous solution [extended ones (70%) and beta-turn (30%)], causes a high degree of flexibility for the backbone.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
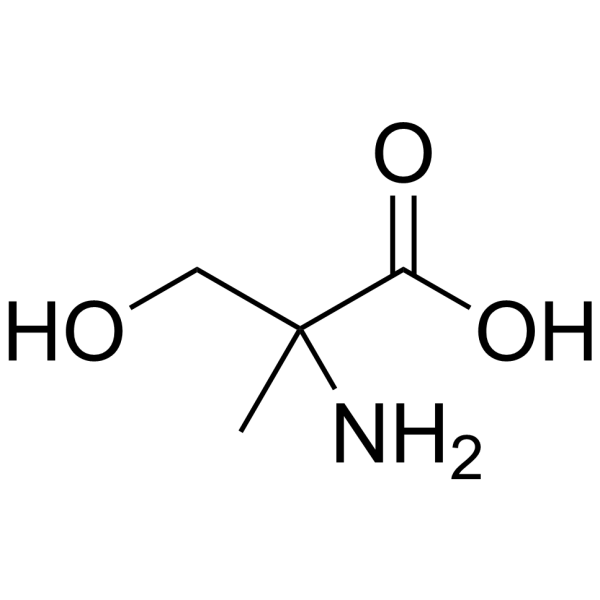 |
2-甲基-DL-丝氨酸
CAS:5424-29-3 |
C4H9NO3 |
|
Drug design, in vitro pharmacology, and structure-activity r...
2009-08-27 [J. Med. Chem. 52 , 5093-107, (2009)] |
|
The requirement of phosphorylation on a threonine residue in...
1993-09-01 [J. Steroid Biochem. Mol. Biol. 46(3) , 337-47, (1993)] |
|
Control of chemoselectivity in Dieckmann ring closures leadi...
2011-10-07 [Org. Biomol. Chem. 9 , 6663, (2011)] |
|
Synthesis, NMR spectra and function of peptides with alpha-m...
1992-10-01 [Acta Chem. Scand. 46(10) , 989-93, (1992)] |
|
Conformational studies on host-guest peptides containing chi...
1988-11-01 [Int. J. Pept. Protein Res. 32(5) , 344-51, (1988)] |