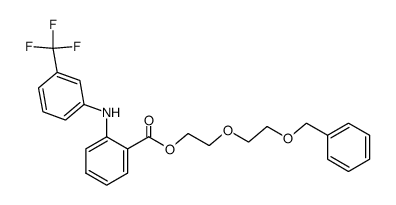
依托芬那酯
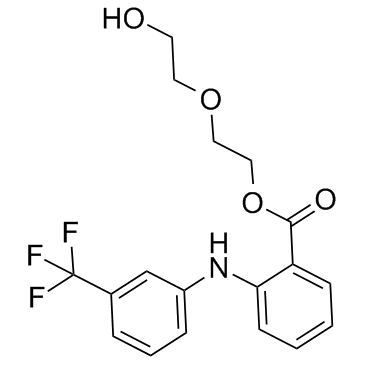
依托芬那酯结构式

|
常用名 | 依托芬那酯 | 英文名 | Etofenamate |
|---|---|---|---|---|
| CAS号 | 30544-47-9 | 分子量 | 369.335 | |
| 密度 | 1.3±0.1 g/cm3 | 沸点 | 451.1±45.0 °C at 760 mmHg | |
| 分子式 | C18H18F3NO4 | 熔点 | 25°C | |
| MSDS | N/A | 闪点 | 226.6±28.7 °C |
依托芬那酯用途Etofenamate是非甾体抗炎化合物。 |
| 中文名 | 依托芬那酯 |
|---|---|
| 英文名 | Etofenamate |
| 中文别名 | 2[[3-(三氟甲基)苯基]氨基]-2-(羟乙氧基)乙基苯甲酸酯 | 2-(2-羟基乙氧基)乙基 2-[3-(三氟甲基)苯胺基]苯甲酸酯 |
| 英文别名 | 更多 |
| 描述 | Etofenamate是非甾体抗炎化合物。 |
|---|---|
| 相关类别 | |
| 参考文献 |
| 密度 | 1.3±0.1 g/cm3 |
|---|---|
| 沸点 | 451.1±45.0 °C at 760 mmHg |
| 熔点 | 25°C |
| 分子式 | C18H18F3NO4 |
| 分子量 | 369.335 |
| 闪点 | 226.6±28.7 °C |
| 精确质量 | 369.118805 |
| PSA | 67.79000 |
| LogP | 4.14 |
| InChIKey | XILVEPYQJIOVNB-UHFFFAOYSA-N |
| SMILES | O=C(OCCOCCO)c1ccccc1Nc1cccc(C(F)(F)F)c1 |
| 外观性状 | light yellow oil |
| 蒸汽压 | 0.0±1.2 mmHg at 25°C |
| 折射率 | 1.552 |
| 储存条件 | Refrigerator |
| 水溶解性 | Practically insoluble in water, miscible with ethanol (96 per cent) and with ethyl acetate. |
| 海关编码 | 2922509090 |
|---|
|
~% 
依托芬那酯 30544-47-9 |
| 文献:Arzneimittel-Forschung/Drug Research, , vol. 27, # 6 b p. 1300 - 1312 |
|
~% 
依托芬那酯 30544-47-9 |
| 文献:Arzneimittel-Forschung/Drug Research, , vol. 27, # 6 b p. 1300 - 1312 |
| 海关编码 | 2922509090 |
|---|---|
| 中文概述 | 2922509090. 其他氨基醇酚、氨基酸酚及其他含氧基氨基化合物. 增值税率:17.0%. 退税率:13.0%. 监管条件:AB. 最惠国关税:6.5%. 普通关税:30.0% |
| 申报要素 | 品名, 成分含量, 用途, 乙醇胺及其盐应报明色度, 乙醇胺及其盐应报明包装 |
| 监管条件 | A.入境货物通关单 B.出境货物通关单 |
| 检验检疫 | R.进口食品卫生监督检验 S.出口食品卫生监督检验 |
| Summary | 2922509090. other amino-alcohol-phenols, amino-acid-phenols and other amino-compounds with oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Adverse reaction of topical etofenamate: petechial eruption.
West Indian Med. J. 61(7) , 767-9, (2012) Etofenamate is a non-steroidal anti-inflammatory drug (NSAID). Clinical findings caused by etofenamate are uncommon. Allergic contact dermatitis is the most common cutaneous reaction reported. But pet... |
|
|
Turpentine sensitization in a nonsteroidal anti-inflammatory solution user.
Dermatitis 23(4) , 182-3, (2012)
|
|
|
Assessing the removal of pharmaceuticals and personal care products in a full-scale activated sludge plant.
Environ. Sci. Pollut. Res. Int. 19(5) , 1818-27, (2012) This study aimed to investigate the removal mechanisms of pharmaceutical active compounds (PhACs) and musks in a wastewater treatment plant (WWTP). Biological removal and adsorption in the activated s... |
| Benzoic acid, 2-[[3-(trifluoromethyl)phenyl]amino]-, 2-(2-hydroxyethoxy)ethyl ester |
| 2-(2-Hydroxyethoxy)ethyl fufenamate |
| 2-[[3-(Trifluoromethyl)phenyl]amino]benzoic Acid 2-(2-Hydroxyethoxy)ethyl Ester |
| Activon |
| 2-(2-hydroxyethoxy)ethyl 2-[3-(trifluoromethyl)anilino]benzoate |
| Bayrogel |
| N-(a,a,a-Trifluoro-m-tolyl)anthranilic Acid 2-(2-Hydroxyethoxy)ethyl Ester |
| UNII-KZF0XM66JC |
| 2-(2-Hydroxyethoxy)ethyl 2-{[3-(trifluoromethyl)phenyl]amino}benzoate |
| Etofenamate |
| Glasel |
| Rheumon |
| Rheumon gel |
| Bayagel |