β-氰基-L-丙氨酸

β-氰基-L-丙氨酸结构式

|
常用名 | β-氰基-L-丙氨酸 | 英文名 | 3-cyano-L-alanine |
|---|---|---|---|---|
| CAS号 | 6232-19-5 | 分子量 | 114.103 | |
| 密度 | 1.3±0.1 g/cm3 | 沸点 | 368.1±32.0 °C at 760 mmHg | |
| 分子式 | C4H6N2O2 | 熔点 | N/A | |
| MSDS | 美版 | 闪点 | 176.4±25.1 °C |
β-氰基-L-丙氨酸用途β-cyano-L-Alanine (Beta-cyano-l-alanine),一种在高等植物中广泛存在的腈,由氰基丙氨酸合成酶从氰化物和半胱氨酸作为底物酶促产生。 β-cyano-L-Alanine 消除了乙醇对脑缺血/再灌注 (I/R) 损伤的保护作用。 |
| 中文名 | β-氰基-L-丙氨酸 |
|---|---|
| 英文名 | 3-cyano-L-alanine |
| 英文别名 | 更多 |
| 描述 | β-cyano-L-Alanine (Beta-cyano-l-alanine),一种在高等植物中广泛存在的腈,由氰基丙氨酸合成酶从氰化物和半胱氨酸作为底物酶促产生。 β-cyano-L-Alanine 消除了乙醇对脑缺血/再灌注 (I/R) 损伤的保护作用。 |
|---|---|
| 相关类别 | |
| 体内研究 | β-氰基-L-丙氨酸(腹腔注射,50 mg/kg)可消除乙醇对脑缺血/再灌注(I/R)损伤的保护作用。动物模型:成年(出生后60天)雄性C57BL/6J小鼠(25-30g)灌胃给予乙醇[2]剂量:50mg/kg给药:腹腔注射结果:取消乙醇对脑I/R损伤的保护作用。 |
| 参考文献 |
| 密度 | 1.3±0.1 g/cm3 |
|---|---|
| 沸点 | 368.1±32.0 °C at 760 mmHg |
| 分子式 | C4H6N2O2 |
| 分子量 | 114.103 |
| 闪点 | 176.4±25.1 °C |
| 精确质量 | 114.042931 |
| PSA | 87.11000 |
| LogP | -0.72 |
| InChIKey | BXRLWGXPSRYJDZ-VKHMYHEASA-N |
| SMILES | N#CCC(N)C(=O)O |
| 蒸汽压 | 0.0±1.8 mmHg at 25°C |
| 折射率 | 1.502 |
| 储存条件 | -20°C |
| 危险品运输编码 | NONH for all modes of transport |
|---|---|
| 海关编码 | 2926909090 |
|
~71% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Arnold,L.D.; May,R.G.; Vederas,J.C. Journal of the American Chemical Society, 1988 , vol. 110, p. 2237 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Phytochemistry (Elsevier), , vol. 29, # 8 p. 2507 - 2508 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Phytochemistry (Elsevier), , vol. 27, # 7 p. 2011 - 2016 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Journal of Organic Chemistry, , vol. 26, p. 3356 - 3360 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:The Journal of organic chemistry, , vol. 36, # 25 p. 3960 - 3966 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Agricultural and Biological Chemistry, , vol. 45, # 9 p. 2053 - 2062 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Agricultural and Biological Chemistry, , vol. 45, # 9 p. 2053 - 2062 |
|
~% 
β-氰基-L-丙氨酸 6232-19-5 |
| 文献:Agricultural and Biological Chemistry, , vol. 45, # 9 p. 2053 - 2062 |
| β-氰基-L-丙氨酸上游产品 10 | |
|---|---|
| β-氰基-L-丙氨酸下游产品 2 | |
| 海关编码 | 2926909090 |
|---|---|
| 中文概述 | 2926909090 其他腈基化合物. 增值税率:17.0% 退税率:9.0% 监管条件:无 最惠国关税:6.5% 普通关税:30.0% |
| 申报要素 | 品名, 成分含量, 用途 |
| Summary | HS:2926909090 other nitrile-function compounds VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:30.0% |
|
Evolution of heteromeric nitrilase complexes in Poaceae with new functions in nitrile metabolism.
Proc. Natl. Acad. Sci. U. S. A. 104(47) , 18848-53, (2007) Members of the nitrilase 4 (NIT4) family of higher plants catalyze the conversion of beta-cyanoalanine to aspartic acid and asparagine, a key step in cyanide detoxification. Grasses (Poaceae) possess ... |
|
|
Vicianin, prunasin, and beta-cyanoalanine in common vetch seed as sources of urinary thiocyanate in the rat.
J. Agric. Food Chem. 49(10) , 5075-80, (2001) When young rats were fed a diet containing common vetch seed for 1 month, they excreted in the urine approximately 7 times more thiocyanate than they had ingested. Vicianin, prunasin, and beta-cyanoal... |
|
|
Maize nitrilases have a dual role in auxin homeostasis and beta-cyanoalanine hydrolysis.
J. Exp. Bot. 58(15-16) , 4225-33, (2007) The auxin indole-3-acetic acid (IAA), which is essential for plant growth and development, is suggested to be synthesized via several redundant pathways. In maize (Zea mays), the nitrilase ZmNIT2 is e... |
| AmbotzHAA5740 |
| 3-cyano-L-alanine |
| b-Cyano-L-Alanine |
| (2S)-2-Amino-3-cyanopropanoic acid |
| L-3-Cyanoalanine |
| β-Cyano-L-alanine |
| (S)-2-amino-3-cyanopropanoic acid |
| L-Alanine,3-cyano |




 CAS号3130-87-8
CAS号3130-87-8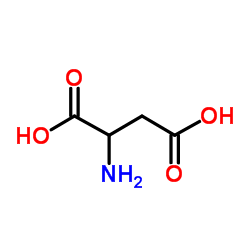 CAS号617-45-8
CAS号617-45-8







