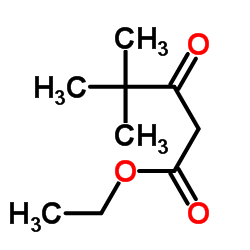
3-叔-丁基-1-苯基-2-吡唑啉-5-酮
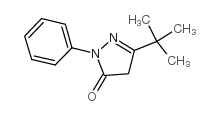
3-叔-丁基-1-苯基-2-吡唑啉-5-酮结构式

|
常用名 | 3-叔-丁基-1-苯基-2-吡唑啉-5-酮 | 英文名 | 3-TERT-BUTYL-1-PHENYL-2-PYRAZOLIN-5-ONE |
|---|---|---|---|---|
| CAS号 | 6631-89-6 | 分子量 | 216.27900 | |
| 密度 | 1.07g/cm3 | 沸点 | 353.6ºC at 760 mmHg | |
| 分子式 | C13H16N2O | 熔点 | 105-108ºC | |
| MSDS | N/A | 闪点 | 167.7ºC |
| 中文名 | 3-叔-丁基-1-苯基-2-吡唑啉-5-酮 |
|---|---|
| 英文名 | 5-tert-butyl-2-phenyl-4H-pyrazol-3-one |
| 英文别名 | 更多 |
| 密度 | 1.07g/cm3 |
|---|---|
| 沸点 | 353.6ºC at 760 mmHg |
| 熔点 | 105-108ºC |
| 分子式 | C13H16N2O |
| 分子量 | 216.27900 |
| 闪点 | 167.7ºC |
| 精确质量 | 216.12600 |
| PSA | 32.67000 |
| LogP | 2.32600 |
| InChIKey | NOSULZKBEJXMKJ-UHFFFAOYSA-N |
| SMILES | CC(C)(C)C1=NN(c2ccccc2)C(=O)C1 |
| 外观性状 | 浅棕色晶体或粉末或霜乳 |
| 折射率 | 1.564 |
| 储存条件 | 室温 |
| 计算化学 | 1.疏水参数计算参考值(XlogP):2.7 2.氢键供体数量:0 3.氢键受体数量:2 4.可旋转化学键数量:2 5.互变异构体数量:3 6.拓扑分子极性表面积32.7 7.重原子数量:16 8.表面电荷:0 9.复杂度:308 10.同位素原子数量:0 11.确定原子立构中心数量:0 12.不确定原子立构中心数量:0 13.确定化学键立构中心数量:0 14.不确定化学键立构中心数量:0 15.共价键单元数量:1 |
| 危害码 (欧洲) | Xi |
|---|---|
| 安全声明 (欧洲) | S22-S24/25 |
| 海关编码 | 2933199090 |
|
~70% 
3-叔-丁基-1-苯基-2-吡... 6631-89-6 |
| 文献:Desroses, Matthieu; Jacques-Cordonnier, Marie-Caroline; Llona-Minguez, Sabin; Jacques, Sylvain; Koolmeister, Tobias; Helleday, Thomas; Scobie, Martin European Journal of Organic Chemistry, 2013 , # 26 p. 5879 - 5885 |
|
~63% 
3-叔-丁基-1-苯基-2-吡... 6631-89-6 |
| 文献:Xu, Xiu-Hua; Wang, Xin; Liu, Guo-Kai; Tokunaga, Etsuko; Shibata, Norio Organic Letters, 2012 , vol. 14, # 10 p. 2544 - 2547 |
|
~74% 
3-叔-丁基-1-苯基-2-吡... 6631-89-6 |
| 文献:Freidmann; Hansel Archiv der Pharmazie, 1983 , vol. 316, # 8 p. 726 - 727 |
| 海关编码 | 2933199090 |
|---|---|
| 中文概述 | 2933199090. 其他结构上有非稠合吡唑环化合物. 增值税率:17.0%. 退税率:13.0%. 监管条件:无. 最惠国关税:6.5%. 普通关税:20.0% |
| 申报要素 | 品名, 成分含量, 用途, 乌洛托品请注明外观, 6-己内酰胺请注明外观, 签约日期 |
| Summary | 2933199090. other compounds containing an unfused pyrazole ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
实验名称:Inhibition of recombinant human LOX expressed in HEK293 cells using diaminopentane as...
来源:ChEMBL
靶标:Protein-lysine 6-oxidase
External Id:CHEMBL4235819
|
|
实验名称:Inhibition of recombinant human LOXL3 expressed in CHO cells using diaminopentane as ...
来源:ChEMBL
靶标:Lysyl oxidase homolog 3
External Id:CHEMBL4235821
|
|
实验名称:Inhibition of recombinant LOXL2 (unknown origin) expressed in NS0 cells using diamino...
来源:ChEMBL
靶标:Lysyl oxidase homolog 2
External Id:CHEMBL4235820
|
|
实验名称:Inhibition of recombinant human LOXL4 expressed in baculovirus infected insect cells ...
来源:ChEMBL
靶标:Lysyl oxidase homolog 4
External Id:CHEMBL4235826
|
|
实验名称:Inhibition of recombinant human LOX expressed in HEK293 cells at 10 uM using diaminop...
来源:ChEMBL
靶标:Protein-lysine 6-oxidase
External Id:CHEMBL4235823
|
|
实验名称:Cell-based high throughput primary assay to identify activators of GPR151
来源:The Scripps Research Institute Molecular Screening Center
靶标:RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id:GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
实验名称:Inhibition of recombinant human LOXL4 expressed in baculovirus infected insect cells ...
来源:ChEMBL
靶标:Lysyl oxidase homolog 4
External Id:CHEMBL4235822
|
|
实验名称:The chemical genetic matrix (CGM) dataset as reported in Wildenhain et al. (2015) Pre...
来源:11924
靶标:N/A
External Id:CGM data for Cell Systems paper Dec 2015
|
|
实验名称:Inhibition of recombinant human LOXL3 expressed in CHO cells at 10 uM using diaminope...
来源:ChEMBL
靶标:Lysyl oxidase homolog 3
External Id:CHEMBL4235825
|
|
实验名称:Inhibition of recombinant LOXL2 (unknown origin) expressed in NS0 cells at 10 uM usin...
来源:ChEMBL
靶标:Lysyl oxidase homolog 2
External Id:CHEMBL4235824
|
| 3H-Pyrazol-3-one,5-(1,1-dimethylethyl)-2,4-dihydro-2-phenyl |
| 5-tert. Butyl-3,4-dihydro-2-phenyl-2H-pyrazol-3-on |
| 3-tert-Butyl-1-phenyl-2-pyrazolin-5-one |
| MFCD00051762 |
| 3-tert-butyl-1-phenyl-1H-pyrazol-5(4H)-one |
| 3-tert-butyl-1-phenyl-2-pyrazol-5-one |
| 5-tert-butyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one |
| 5-tert-butyl-2-phenyl-2,4-dihydropyrazol-3-one |
| 3-tert-butyl-1-phenyl-5(1H)-pyrazolone |