| Structure | Name/CAS No. | Articles |
|---|---|---|
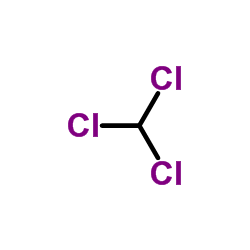 |
Chloroform
CAS:67-66-3 |
|
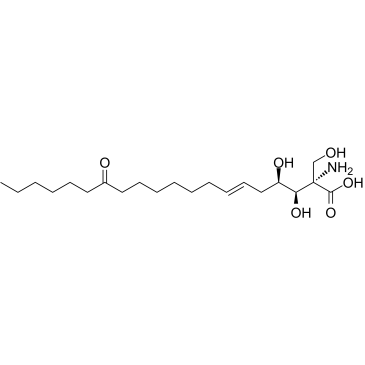 |
Myriocin
CAS:35891-70-4 |
|
 |
ethyl acetate
CAS:141-78-6 |
|
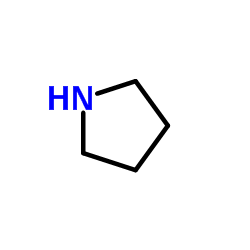 |
Pyrrolidine
CAS:123-75-1 |