Chemistry: A European Journal
2008-01-01
Asymmetric synthesis of (+)-altholactone: a styryllactone isolated from various Goniothalamus species.
Dieter Enders, Julien Barbion
Index: Chemistry 14(9) , 2842-9, (2008)
Full Text: HTML
Abstract
The asymmetric total synthesis of (+)-altholactone (1), a member of the styryllactone family of natural products displaying cytotoxic and antitumor activities, is described. Key steps include a RAMP-hydrazone alpha-alkylation (RAMP=(R)-1-amino-2-methoxymethylpyrrolidine) of 2,2-dimethyl-1,3-dioxan-5-one, a boron-mediated aldol reaction, a six- to five-membered ring acetonide shuffling, an oxidative 1,5-diol to delta-lactone conversion and a stereoselective ring-closure to generate the annulated tetrahydrofuran moiety with inversion of configuration.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
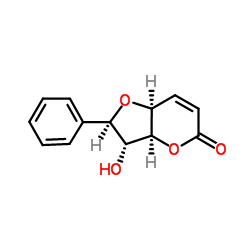 |
Altholactone
CAS:65408-91-5 |
C13H12O4 |
Related Articles:
More...
|
Stereoselective total synthesis of bioactive styryllactones ...
2008-01-04 [J. Org. Chem. 73(1) , 2-11, (2008)] |
|
The cytotoxicity of naturally occurring styryl lactones.
2006-02-01 [Phytomedicine 13(3) , 181-6, (2006)] |
|
Stereospecificity in the Au-catalysed cyclisation of monoall...
2011-07-21 [Chem. Commun. (Camb.) 47(27) , 7659-61, (2011)] |
|
A convergent Pd-catalyzed asymmetric allylic alkylation of d...
2007-01-01 [Chemistry 13(34) , 9547-60, (2007)] |
|
Altholactone induces apoptotic cell death in human colorecta...
2012-06-01 [Phytother Res. 26(6) , 926-31, (2012)] |