Altholactone
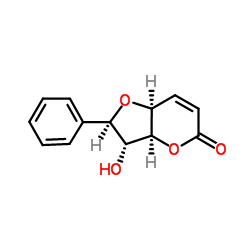
Altholactone structure
|
Common Name | Altholactone | ||
|---|---|---|---|---|
| CAS Number | 65408-91-5 | Molecular Weight | 232.23 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 490.4±45.0 °C at 760 mmHg | |
| Molecular Formula | C13H12O4 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 196.1±22.2 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
|
Stereoselective total synthesis of bioactive styryllactones (+)-goniofufurone, (+)7-epi-goniofufurone, (+)-goniopypyrone, (+)-goniotriol, (+)-altholactone, and (-)-etharvensin.
J. Org. Chem. 73(1) , 2-11, (2008) Stereoselective total synthesis of biologically active styryllactones 7-epi-goniofufurone, goniofufurone, goniopypyrone, goniotriol, altholactone, and etharvensin was achieved in high overall yields from a common intermediate derived from d-(-)-tartaric acid.... |
|
|
The cytotoxicity of naturally occurring styryl lactones.
Phytomedicine 13(3) , 181-6, (2006) We extracted and isolated three natural styryl lactones from Goniothalamus griffithii Hook f. Thoms and investigated their cytotoxicity on a panel of three hepatocyte cell lines, HepG2, drug resistant HepG2 (HepG2-R) and primary cultured normal mice hepatocyt... |
|
|
Asymmetric synthesis of (+)-altholactone: a styryllactone isolated from various Goniothalamus species.
Chemistry 14(9) , 2842-9, (2008) The asymmetric total synthesis of (+)-altholactone (1), a member of the styryllactone family of natural products displaying cytotoxic and antitumor activities, is described. Key steps include a RAMP-hydrazone alpha-alkylation (RAMP=(R)-1-amino-2-methoxymethyl... |
|
|
Stereospecificity in the Au-catalysed cyclisation of monoallylic diols. Synthesis of (+)-isoaltholactone.
Chem. Commun. (Camb.) 47(27) , 7659-61, (2011) We describe a concise synthesis of (+)-isoaltholactone via a Au-catalysed cyclisation of a monoallylic diol to form the tetrahydrofuranyl ring. Analogous cyclisations show that the stereochemical outcome is dictated by the stereochemistry of the diol substrat... |
|
|
A convergent Pd-catalyzed asymmetric allylic alkylation of dl- and meso-divinylethylene carbonate: enantioselective synthesis of (+)-australine hydrochloride and formal synthesis of isoaltholactone.
Chemistry 13(34) , 9547-60, (2007) The use of a mixture of dl- and meso-divinylethylene carbonate as an electrophile in palladium-catalyzed asymmetric allylic alkylation reactions is reported. From the diastereomeric mixture of meso and chiral racemic starting materials, a single product is ob... |
|
|
Altholactone induces apoptotic cell death in human colorectal cancer cells.
Phytother Res. 26(6) , 926-31, (2012) Resistance of colorectal cancer (CRC) to the available chemotherapy reveals the demand for identification of new anticancer agents. We evaluated the antitumour potential of altholactone, a naturally occurring bioactive compound isolated from Goniothalamus spp... |
|
|
Apoptotic activities in closely related styryllactone stereoisomers toward human tumor cell lines: Investigation of synergism of styryllactone-induced apoptosis with TRAIL.
Bioorg. Med. Chem. 18(2) , 849-54, (2010) A related series of styryllactones with small functional and stereochemical variations were compiled for a comparative study of their apoptotic activities toward two tumorigenic and one non-tumorigenic control cell line. While a substantial range of intrinsic... |
|
|
Altholactone displays promising antimicrobial activity.
Molecules 16(6) , 4560-6, (2011) The antimicrobial activity of altholactone, a naturally extracted styryllactone isolated from Goniothalamus malayanus, was determined against Gram positive (S. aureus ATTC 25923, S. aureus ATTC 25392, and E. faecalis ATTC 29212) and Gram negative (E. coli ATT... |