| Structure | Name/CAS No. | Articles |
|---|---|---|
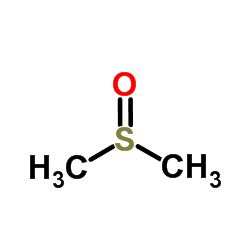 |
Dimethyl sulfoxide
CAS:67-68-5 |
|
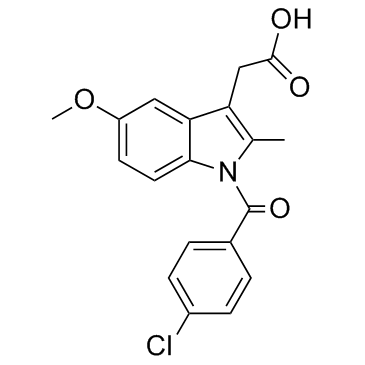 |
Indometacin
CAS:53-86-1 |
|
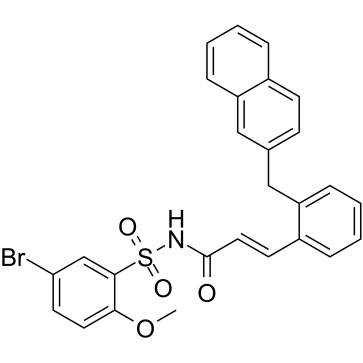 |
L-798106
CAS:244101-02-8 |
|
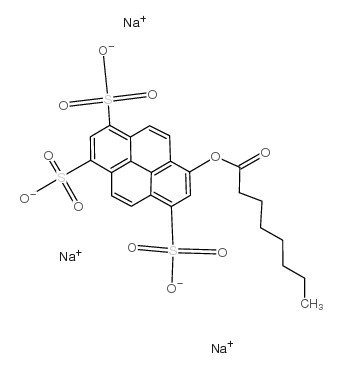 |
8-Octanoyloxypyrene-1,3,6-trisulfonic acid trisodium salt
CAS:115787-84-3 |