| Structure | Name/CAS No. | Articles |
|---|---|---|
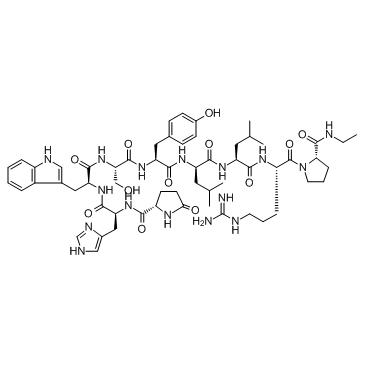 |
Leuprolide TFA
CAS:53714-56-0 |
|
![[D-Trp6]-LH-RH Structure](https://image.chemsrc.com/caspic/100/57773-63-4.png) |
[D-Trp6]-LH-RH
CAS:57773-63-4 |
|
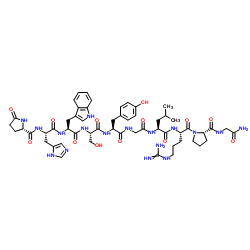 |
Gonadorelin
CAS:33515-09-2 |
|
 |
(Des-Gly10,D-Leu6,Pro-NHEt9)-LHRH acetate salt
CAS:74381-53-6 |