| Structure | Name/CAS No. | Articles |
|---|---|---|
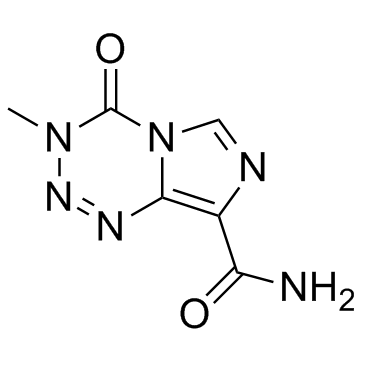 |
Temozolomide
CAS:85622-93-1 |
|
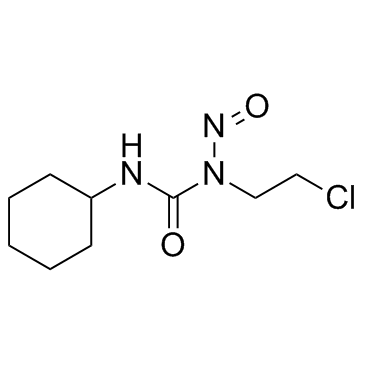 |
Lomustine
CAS:13010-47-4 |