Stereoselective rearrangement of trichloroacetimidates: application to the synthesis of alpha-glycosyl ureas.
Yothin Teethaisong, Nongluk Autarkool, Kittipot Sirichaiwetchakoon, Pongrit Krubphachaya, Sajeera Kupittayanant, Griangsak Eumkeb
Index: Org. Lett. 11(11) , 2433-6, (2009)
Full Text: HTML
Abstract
A new method for the stereoselective synthesis of alpha-glycosyl ureas, via nickel-catalyzed [1,3]-rearrangement of glycosyl trichloroacetimidates, has been developed. The alpha-stereoselectivity at the anomeric carbon of the resulting trichloroacetamides depends on the nature of the cationic nickel catalyst. This method is applicable to a number of trichloroacetimidate substrates. The alpha-glycosyl trichloroacetamides can be directly converted into alpha-glycosyl ureas in the presence of amines. In all cases, the stereochemical integrity at the urea linkages remains intact.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
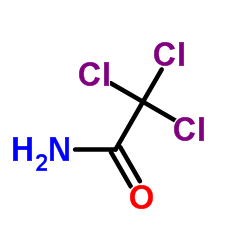 |
2,2,2-Trichloroacetamide
CAS:594-65-0 |
C2H2Cl3NO |
|
An improved synthesis of (-)-5,11-dideoxytetrodotoxin.
2013-02-15 [J. Org. Chem. 78(4) , 1699-705, (2013)] |
|
Activation of glycosyl trichloroacetimidates with perchloric...
2010-09-23 [Carbohydr. Res. 345(14) , 2074-8, (2010)] |
|
Synthetic studies toward the anthrax tetrasaccharide: altern...
2012-07-15 [Carbohydr. Res. 356 , 115-31, (2012)] |
|
Rhodium-catalyzed regio- and enantioselective amination of r...
2012-12-07 [Chem. Commun. (Camb.) 48(94) , 11531-3, (2012)] |
|
Influence of the solvent in low temperature glycosylations w...
2011-09-06 [Carbohydr. Res. 346(12) , 1495-502, (2011)] |