[Lipase-catalyzed enantioselective ammonolysis of racemic phenylglycine methyl ester in organic solvent].
Wei Du, Min-Hua Zong, Yong Guo, Jun He, Yuan-Yuan Zhang, Zhao-Lin Xie, Wen-Yong Lou
Index: Sheng Wu Gong Cheng Xue Bao 18(2) , 242-5, (2002)
Full Text: HTML
Abstract
A novel reaction-enzymatic ammonolysis discovered in the mid of 1990s has been demonstrated to be a very promising alternative in the preparation of optically pure compounds. The effects of organic solvent, initial water activity, temperature and additives on lipase Novozym435-catalyzed enantioselective ammonolysis of racemic phenylglycine methyl ester were investigated systematically in this paper. Enzymatic reaction of ammonolysis showed higher activity and enantioselectivity than the corresponding reaction of hydrolysis and alcoholysis.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
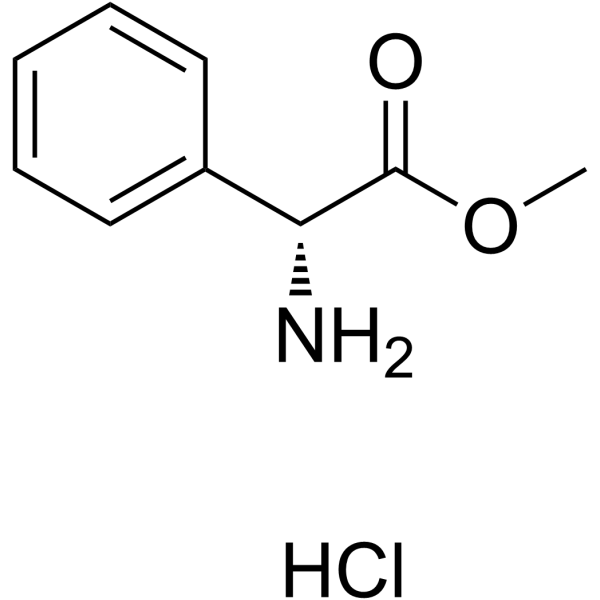 |
H-D-Phg-OMe.HCl
CAS:19883-41-1 |
C9H12ClNO2 |
|
Improving lipase-catalyzed enantioselective ammonolysis of p...
2003-03-01 [Biotechnol. Lett. 25(6) , 461-4, (2003)] |
|
Lipase-catalysed enantioselective ammonolysis of phenylglyci...
2003-10-01 [Biotechnol. Appl. Biochem. 38(Pt 2) , 107-10, (2003)] |
|
Kinetics of ampicillin synthesis catalyzed by penicillin acy...
2000-12-01 [Biochemistry. (Mosc.) 65(12) , 1367-75, (2000)] |
|
Enhanced enzymatic synthesis of a semi-synthetic cephalospri...
2003-07-01 [Biotechnol. Lett. 25(14) , 1195-8, (2003)] |
|
Life cycle assessment of a coupled solar photocatalytic-biol...
2006-11-01 [Water Res. 40(19) , 3533-40, (2006)] |