The myosin ATPase inhibitor, 2,3-butanedione 2-monoxime, prevents protein secretion by the basidiomycete Coprinopsis cinerea.
Kohsuke Hashimoto, Etsuo Yokota, Teruo Shimmen, Makoto Yoshida
Index: Biotechnol. Lett. 33(4) , 769-75, (2011)
Full Text: HTML
Abstract
The plant-saprophytic basidiomycete, Coprinopsis cinerea, produces and secretes various cellulases during cellulose degradation as the main extracellular proteins. Although enzymatic characterization of such cellulases has been frequently reported, the mechanism of their secretion remains unclear. This study focused on myosins, actin-based motor proteins, involved in protein secretion in C. cinerea. During cultivation under cellulase-inducing condition, no cellulase activity was observed when the mycelia were treated with 2,3-butanedione 2-monoxime (BDM), a general inhibitor of myosin ATPase. Furthermore, BDM treatment disrupted the localization of the Golgi apparatus, but not that of the endoplasmic reticulum. Three genes encoding myosin-like proteins (CcMyo1, CcMyo2 and CcMyo5) were identified from the C. cinerea genome database. Transcription of these genes was promoted when the fungus was grown under cellulase-inducing condition.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
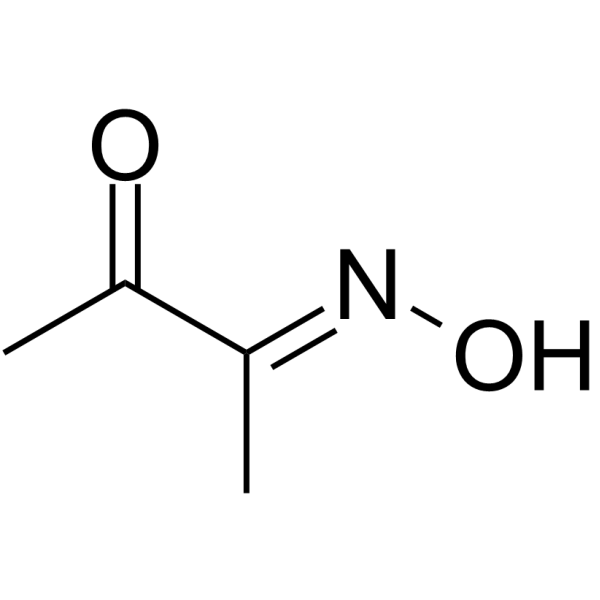 |
Diacetyl monoxime
CAS:57-71-6 |
C4H7NO2 |
|
Carbamylation of immunoglobulin abrogates activation of the ...
2014-11-01 [Eur. J. Immunol. 44(11) , 3403-12, (2014)] |
|
Elastic proteins in the flight muscle of Manduca sexta.
2015-02-15 [Arch. Biochem. Biophys. 568 , 16-27, (2015)] |
|
Metrics and clinical relevance of percutaneous penetration a...
2015-01-01 [Skin Pharmacol. Physiol. 28(2) , 57-64, (2015)] |
|
Mitochondria-targeted ROS scavenger improves post-ischemic r...
2014-12-01 [J. Mol. Cell. Cardiol. 77 , 136-46, (2015)] |
|
Cutaneous challenge with chemical warfare agents in the SKH-...
2011-06-01 [Hum. Exp. Toxicol. 30(6) , 491-8, (2011)] |