Chemistry & Biodiversity
2004-01-01
One-step synthesis of hypoxanthine from glycinamide and diformylurea.
Irene M Lagoja, Piet Herdewijn
Index: Chem. Biodivers. 1(1) , 106-11, (2004)
Full Text: HTML
Abstract
Because of their easy availability and their relative chemical stability, urea, formic acid, and glycine might have played a role in the assembly process of nucleobases. In this paper, a short reaction path is described to prepare hypoxanthine starting from the above mentioned precursors. The formation of hypoxanthine has been verified by high-resolution mass spectrometry with the 15N-labelled urea as starting material, and HPLC analysis. The yield of this condensation reaction has been determined spectrophotometrically.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
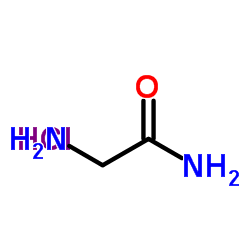 |
H-Gly-NH2.HCl
CAS:1668-10-6 |
C2H7ClN2O |
Related Articles:
More...
|
Preparation of guanine and diaminopurine from biuret. Part I...
2007-04-01 [Chem. Biodivers. 4(4) , 818-22, (2007)] |
|
Identification of quinoline, carboline and glycinamide compo...
2013-12-01 [Food Chem. 141(3) , 1888-94, (2013)] |
|
Synthesis of (-)-quinocarcin by directed condensation of alp...
2005-12-07 [J. Am. Chem. Soc. 127(48) , 16796-7, (2005)] |
|
Clozapine and glycinamide prevent MK-801-induced deficits in...
2014-09-01 [Behav. Brain Res. 271 , 203-11, (2014)] |
|
Design and characterization of a noncompetitive antagonist o...
2006-10-01 [J. Pain 7(10) , 735-46, (2006)] |