90141-22-3
| Name | (R)-(-)-Gossypol |
|---|---|
| Synonyms |
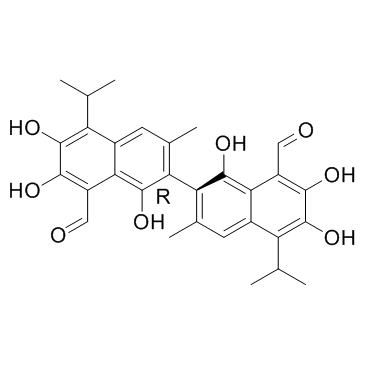
1,1',6,6',7,7'-Hexahydroxy-5,5'-diisopropyl-3,3'-dimethyl[2,2'-binaphthalene]-8,8'-dicarboxaldehyde
UNII:XNA7DR63CQ 1,1',6,6',7,7'-Hexahydroxy-5,5'-diisopropyl-3,3'-dimethyl-2,2'-binaphthalene-8,8'-dicarbaldehyde UNII:8DY2X8LXW4 2,2'-Bis[1,6,7-trihydroxy-3-methyl-5-isopropyl-8-aldehydonaphthalene] 1,1',6,6',7,7'-hexahydroxy-3,3'-dimethyl-5,5'-di(propan-2-yl)-2,2'-binaphthalene-8,8'-dicarbaldehyde Gossypol UNII:KAV15B369O 2,2'-bis[8-formyl-1,6,7-trihydroxy-5-isopropyl-3-methylnaphthalene] (-)-1,1',6,6',7,7'-Hexahydroxy-3,3'-dimethyl-5,5'-bis(1-methylethyl)[2,2'-binaphthalene]-8,8'-dicarboxaldehyde 1,1',6,6',7,7'-Hexahydroxy-5,5'-diisopropyl-3,3'-dimethyl-2,2'-binaphthalen-8,8'-dicarbaldehyd 1,1',6,6',7,7'-hexahydroxy-3,3'-dimethyl-5,5'-bis(1-methylethyl)-2,2'-binaphthalene-8,8'-dicarbaldehyde 1,1',6,6',7,7'-Hexahydroxy-3,3'-dimethyl-5,5'-bis(1-methylethyl)[2,2'-binaphthalene]-8,8'-dicarboxaldehyde AT-101 |
| Description | (R)-(-)-Gossypol (AT-101) is the levorotatory isomer of a natural product Gossypol. AT-101 is determined to bind to Bcl-2, Mcl-1 and Bcl-xL proteins with Kis of 260±30 nM, 170±10 nM, and 480±40 nM, respectively. |
|---|---|
| Related Catalog | |
| Target |
Bcl-2:260 nM (Ki) Bcl-xL:480 nM (Ki) Mcl-1:170 nM (Ki) Autophagy |
| In Vitro | The natural racemic Gossypol has two enantiomers, namely the (R)-(-)-Gossypol (AT-101) and (+)-Gossypol enantiomers. (R)-(-)-Gossypol (AT-101) and (+)-Gossypol binds to Bcl-2 or Bcl-xL with similar binding affinities, AT-101 is more potent than (+)-Gossypol in inhibition of cell growth and induction of apoptosis, possibly due to the influence of serum in the cell culture experiments. The racemic form and each of the enantiomers of Gossypol are tested against UM-SCC-6 and UM-SCC-14A in 6-day MTT assays. AT-101 exhibits greater growth inhibition relative to (±)-Gossypol than (+)-Gossypol in both cell lines tested (P<0.001). An intermediate growth inhibitory effect is observed with (±)-Gossypol but this effect is only observed at the higher dose of Gossypol (10 μM, P<0.0001). (R)-(-)-Gossypol (AT-101) binds to the BH3-binding groove of Bcl-xL and Bcl-2 proteins with fairly high affinity, has potent activity against head and neck squamous cell carcinomas (HNSCC) cell lines in vitro. Furthermore, it induces apoptosis with high efficiency in HNSCC tumor cells that express functional p53 and that also kills tumor cells with mutant p53 by a different mechanism. AT-101 doses required to inhibit the growth of human fibroblast cell lines by 50% were 2- to 10-fold higher than for HNSCC cell lines. To inhibit human oral keratinocyte growth by 50%, (R)-(-)-Gossypol (AT-101) concentrations are 2-to 3-fold higher than for HNSCC cell lines. (R)-(-)-Gossypol (AT-101) causes dose-dependent inhibition of cell growth in ten UM-SCC cell lines over a range from 0.5 to 10 μM in a 6-day MTT assay. The relative sensitivity of the cell lines vary from a very sensitive group with an IC50 of 2-5 μM and a less sensitive group with IC50 clusters around 10 μM[1]. (R)-(-)-Gossypol (AT-101) is determined to bind to Bcl-2, Mcl-1 and Bcl-xL proteins with Ki values of 260±30 nM, 170±10 nM, and 480±40 nM, respectively[2]. |
| Cell Assay | Two representative UM-SCC cell lines, UM-SCC-6 and UM-SCC-14A, are continuously exposed to 0 (vehicle control), 5 or 10 μM (±)-Gossypol, (R)-(-)-Gossypol or (+)-Gossypol in a 6-day MTT cell survival assay[1]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 707.9±55.0 °C at 760 mmHg |
| Melting Point | 166-167ºC |
| Molecular Formula | C30H30O8 |
| Molecular Weight | 518.554 |
| Flash Point | 395.9±28.0 °C |
| Exact Mass | 518.194092 |
| PSA | 155.52000 |
| LogP | 6.16 |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.742 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|