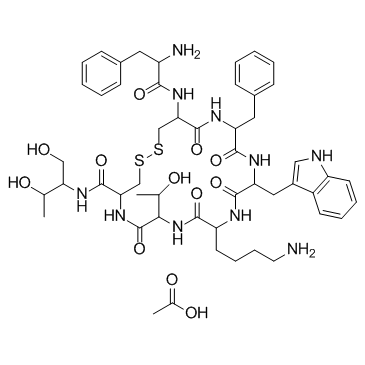
79517-01-4
| Name | acetic acid,10-(4-aminobutyl)-19-[(2-amino-3-phenylpropanoyl)amino]-16-benzyl-N-(1,3-dihydroxybutan-2-yl)-7-(1-hydroxyethyl)-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carboxamide |
|---|---|
| Synonyms |
Sandostatin (TN)
Octreotide acetate (USAN) 1,2-Dithia-5,8,11,14,17-pentaazacycloeicosane-4-carboxamide, 10-(4-aminobutyl)-19-[[(2R)-2-amino-1-oxo-3-phenylpropyl]amino]-7-(1-hydroxyethyl)-N-[(1R,2R)-2-hydroxy-1-(hydroxymethyl)propyl]-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-16-(phenylmethyl)-, (4R,7S,10S,13R,16S,19R)-, acetate (1:1) (salt) Octreotide (acetate) (4R,7S,10S,13R,16S,19R)-10-(4-Aminobutyl)-16-benzyl-N-[(2R,3R)-1,3-dihydroxy-2-butanyl]-7-(1-hydroxyethyl)-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-19-(D-phenylalanylamino)-1,2-dithia-5,8,11,14,17-pentaazacycloicosane-4-carboxamide acetate (1:1) Octreotide Octreotide Acetate SMS 201-995 Sandostatin LAR (4R,7S,10S,13R,16S,19R)-10-(4-Aminobutyl)-16-benzyl-N-[(2R,3R)-1,3-dihydroxy-2-butanyl]-7-[(1R)-1-hydroxyethyl]-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-19-(D-phenylalanylamino)-1,2-dithia-5,8,11,14,17-pentaazacycloicosane-4-carboxamide Sandostatin (4R,7S,10S,13R,16S,19R)-10-(4-aminobutyl)-16-benzyl-N-[(2R,3R)-1,3-dihydroxybutan-2-yl]-7-(1-hydroxyethyl)-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-19-(D-phenylalanylamino)-1,2-dithia-5,8,11,14,17-pentaazacycloicosane-4-carboxamide acetate (1:1) zacycloicosane-4-carboxamide acetate |
| Description | Octreotide acetate, a long-acting synthetic analog of native somatostatin, inhibits growth hormone, glucagon, and insulin more potently. |
|---|---|
| Related Catalog | |
| In Vivo | Octreotide-treated groups show a significant reduction in the tumor volume when compared with saline group. Octreotide-PPSG (1.4 mg/kg, i.p.) shows greater antitumor effect than Octreotide-soln (100 μg/kg, i.p.). Octreotide-treatments results in significant inhibitory effect on the expression levels of SSTR2 and SSTR5 in primary HCC-bearing rats compared with the saline group. Octreotide-PPSG appears to inhibit the expression of SSTR2 and SSTR5 to a greater extent than that of Octreotide-soln treated group[1]. A test dose of octreotide acetate significantly decreases the serum gastrin level to approximately one third of the baseline in 2 hr and the effect lasted approximately for 6 hr. On day 21, treatment with sustained-release formulation of octreotide acetatea (5 mg intramuscular, q 4 wk) is initiated[2]. |
| Animal Admin | Mice[1] Thirty mice with HCC xenografts are randomLy divided into three groups: (A) Octreotide-soln group, (B) Octreotide-PPSG group, and (C) control group. Octreotide-soln group receives i.p. injection of 100 μg/kg octreotide-soln once a day and totally for consecutive 14 days. Octreotide-PPSG group receives a single subcutaneous injection of 1.4 mg/kg Octreotide-PPSG, and the injection volume is about 0.2 mL. Control group receives i.p. injection of saline once a day for consecutive 14 days. Treatment starts on the next day after injection of H22 hepatoma cell suspension and maintains for 14 days. Tumor growth is monitored by periodic caliper measurements on day 7 and day 14 post seeding. Tumor volumes (V) are calculated based on the length and width of tumor by Eq. Rats[1] Twelve male SD rats are divided into two groups, and housed in standard cages at 25°C, with free access to food and water for a week prior to the experiment. Rats are subcutaneously injected with Octreotidereotide solution (Octreotide-soln) or Octreotide-PPSG at an equivalent single dose of 20 mg/kg. The dose is determined based on the clinical dose of Octreotide-soln in human. Rats are fasted for 12 h before dosing and food is returned approximately 2 h post dosing. Blood samples are collected at predetermined time points using heparinized Eppendorf tubes. Immediately after collection, the blood samples are placed on ice until centrifuged at 3000 g for 10 min within 1 h. The plasma is collected and stored at −20°C until analysis. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 1447.2±65.0 °C at 760 mmHg |
| Melting Point | 153-156ºC |
| Molecular Formula | C51H70N10O12S2 |
| Molecular Weight | 1079.29 |
| Flash Point | 829.1±34.3 °C |
| PSA | 382.82000 |
| LogP | 0.77 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.673 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|---|
| WGK Germany | 3 |