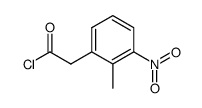
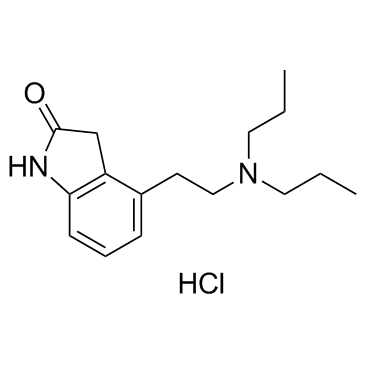
91374-21-9
| Name | ropinirole |
|---|---|
| Synonyms |
Ropinirole
Unii-030pyr8953 MFCD00864147 Ropinirolum 4-[2-(Dipropylamino)ethyl]-1,3-dihydro-2H-indol-2-one ROPINIROLE INTERMEIDATES: |
| Description | Ropinirole (SKF 101468) is an orally active, potent D3/D2 receptor agonist with a Ki of 29 nM for D2 receptor. Ropinirole has pEC50s of 7.4, 8.4 and 6.8 for hD2, hD3 and hD4 receptors, respectively. Ropinirole has no affinity for the D1 receptors. Ropinirole has the potential for Parkinson's disease[1][2]. |
|---|---|
| Related Catalog | |
| Target |
D2 Receptor:29 nM (Ki) hD2 Receptor:7.4 (pEC50) hD3 Receptor:8.4 (pEC50) hD4.4 Receptor:6.8 (pEC50) |
| In Vitro | Ropinirole has affinity for D3 receptors of 10-20 fold higher than the D2 and D4 receptors. Ropinirole is weakly active at alpha 2-adrenoceptors and 5-HT2 receptors but inactive at 5-HT1, benzodiazepine and gamma-aminobutyric acid receptors or alpha 1 and beta-adrenoceptors[1][2]. |
| In Vivo | Ropinirole (0.1-10 mg/kg; i.p.) decreases intracranial self-stimulation (ICSS) thresholds and induces anxiolytic- and antidepressive-like effects without affecting motor activity or spatial memory[2]. |
| References |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 410.5±45.0 °C at 760 mmHg |
| Melting Point | 243-250°C |
| Molecular Formula | C16H17D7N2O |
| Molecular Weight | 260.375 |
| Flash Point | 202.0±28.7 °C |
| Exact Mass | 260.188873 |
| PSA | 32.34000 |
| LogP | 3.19 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.539 |
| Hazard Codes | Xi |
|---|---|
| Risk Phrases | R36/37/38:Irritating to eyes, respiratory system and skin . |
| Safety Phrases | S26-S36/37/39 |
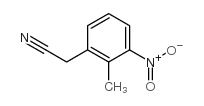
| Precursor 10 | |
|---|---|
| DownStream 1 | |
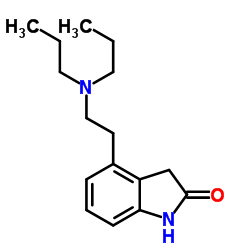
![N-[2-(1H-indol-4-yl)ethyl]-N-propylpropan-1-amine structure](https://image.chemsrc.com/caspic/461/76149-15-0.png)

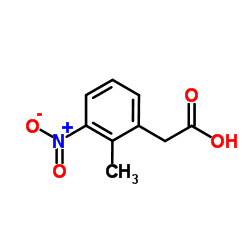
![Benzeneacetic acid,2-[2-(dipropylamino)ethyl]-6-nitro structure](https://image.chemsrc.com/caspic/248/720656-64-4.png)
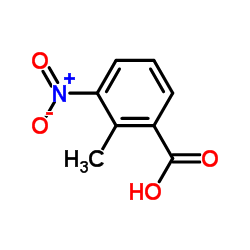

![4-[2-(dipropylamino)ethyl]-7-hydroxy-1,3-dihydroindol-2-one structure](https://image.chemsrc.com/caspic/268/81654-62-8.png)