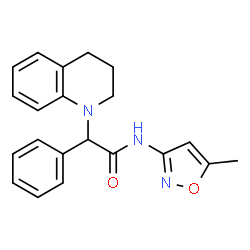
1031496-06-6
| Name | CIM0216 |
|---|---|
| Synonyms |
2-(3,4-Dihydro-1(2H)-quinolinyl)-N-(5-methyl-1,2-oxazol-3-yl)-2-phenylacetamide
MFCD10627035 CIM0216 |
| Description | CIM0216, a potent and selective agonist of TRPM3, has the ability to open two distinct cation-permeable pores in TRPM3. CIM0216 exhibits selectivity for TRPM3 over TRPM1, TRPM2 and TRPM4-8. CIM0216 acts as a powerful tool for use in investigating the physiological roles of TRPM3, and can be used for neurogenic inflammation research[1]. |
|---|---|
| Related Catalog | |
| Target |
TRPM3 |
| In Vitro | CIM0216 elicits a dose-dependent Ca2+ response [pEC50=0.77±0.1 µM] in HEK-TRPM3 cells, which is not observed in nontransfected HEK293 cells. CIM0216 induces a robust increase in intracellular Ca2+ concentration (1,145±26 nM) in single-cell FURA2-ratiometric Ca2+ imaging in HEK-TRPM3 cells. These responses are not observed in nontransfected HEK cells or in the absence of extracellular Ca2+[1]. CIM0216 (10 µM) has no stimulating/blocking effect on TRPM1, TRPM4, TRPM6, or TRPM7 currents; however, a small blocking effect of CIM0216 is observed after activation of TRPM2 (16.6% block) and TRPM5 (33.5% block). CIM0216 also has no detectable effect on human TRPV1 and TRPM8 channel activation[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 608.7±55.0 °C at 760 mmHg |
| Molecular Formula | C21H21N3O2 |
| Molecular Weight | 347.410 |
| Flash Point | 321.9±31.5 °C |
| Exact Mass | 347.163391 |
| LogP | 3.18 |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.646 |
| Storage condition | -20°C |