152095-12-0
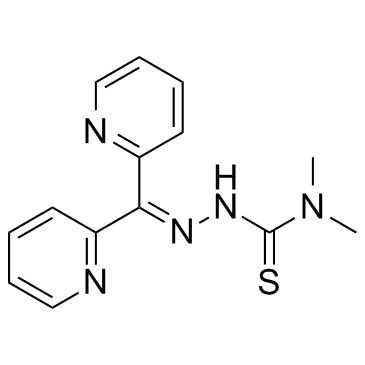
| Name | 3-(dipyridin-2-ylmethylideneamino)-1,1-dimethylthiourea |
|---|---|
| Synonyms |
2-(Di-2-pyridinylmethylene)-N,N-dimethylhydrazinecarbothioamide
2-(dipyridin-2-ylmethylidene)-N,N-dimethylhydrazinecarbothioamide Hydrazinecarbothioamide,2-(di-2-pyridinylmethylene)-N,N-dimethyl Iron Chelator,Dp44mT 2,N-dimethylsemicarbazone Dp44mT |
| Description | Dp44mT is an iron chelator with selective anticancer activity. |
|---|---|
| Related Catalog | |
| Target |
Target: Iron chelator[1] |
| In Vitro | Dp44mT is cytotoxic to breast cancer cells, at least in part, due to selective inhibition of top2α. Dp44mT alone induced selective cell killing in the breast cancer cell line MDA-MB-231 when compared with healthy mammary epithelial cells (MCF-12A). It induces G1 cell cycle arrest and reduces cancer cell clonogenic growth at nanomolar concentrations. Dp44mT, but not the iron chelator desferal, induces DNA double-strand breaks quantified as S139 phosphorylated histone foci (γ-H2AX) and Comet tails induced in MDA-MB-231 cells. Doxorubicin-induced cytotoxicity and DNA damage are both enhanced significantly in the presence of low concentrations of Dp44mT. The chelator caused selective poisoning of DNA topoisomerase IIα (top2α) as measured by an in vitro DNA cleavage assay and cellular topoisomerase-DNA complex formation[1]. Dp44mT targets lysosome integrity through copper binding. Copper binding is essential for the potent antitumor activity of Dp44mT, as coincubation with nontoxic copper chelators markedly attenuated its cytotoxicity[2]. |
| Kinase Assay | For DNA topoisomerase IIα assays, the 161-bp fragment from pBluescript SK(-) phagemid DNA or single stranded oligonucleotides are 5'-end labeled with [32P]ATP and T4 polynucleotide kinase. Labeling mixtures are subsequently centrifuged through Mini Quick Spin DNA columns (for pSK fragments) or Oligo columns (for oligonucleotides) to remove the unincorporated label. Annealing to the complementary strand of the oligonucleotides is done by heating the reaction mixture to 95°C and overnight cooling to room temperature in 10 mM Tris-HCl (pH 7.8), 100 mM NaCl, and 1 mM EDTA. DNA substrates (10 pmol/reaction) are incubated with 500 ng of top2a or top2h in the presence or absence of Dp44mT for the indicated times at 25°C in 10 μL of reaction buffer. Reactions are stopped by adding SDS (final concentration 0.5%). Samples are separated on 16% (for pSK DNA) or 20% (for the oligonucleotides) denaturing polyacrylamide gels (7 M urea). Imaging and quantitation are done using a PhosphorImager[1]. |
| Cell Assay | Cell proliferation is measured using a sulforhodamine B dye–based assay. MDA-MB-231(breast cancer) and MCF-12A (healthy mammary epithelial) cells are incubated with increasing concentrations of Dp44mT (0.01, 0.1, 1, 10, 100 μM). Results are expressed relative to control[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 438.4±43.0 °C at 760 mmHg |
| Molecular Formula | C14H15N5S |
| Molecular Weight | 285.367 |
| Flash Point | 218.9±28.2 °C |
| Exact Mass | 285.104828 |
| PSA | 92.54000 |
| LogP | 1.21 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.635 |
| Storage condition | -20℃ |
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 |
| RIDADR | UN 2811 6.1 / PGIII |