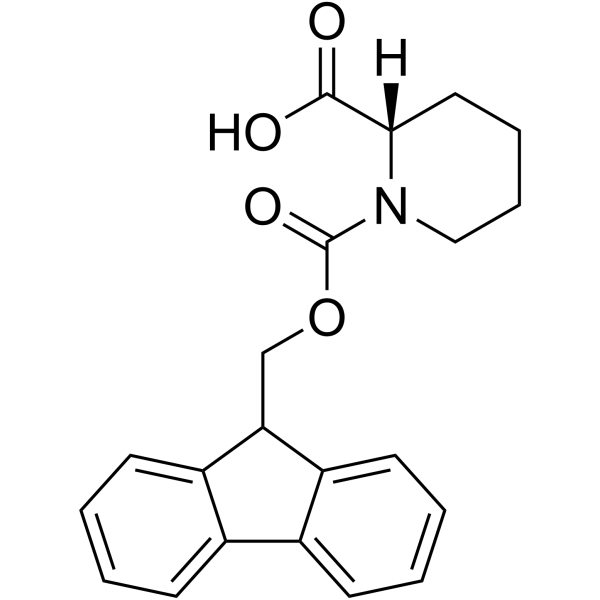
86069-86-5
| Name | Fmoc-(S)-(-)-piperidine-2-carboxylic acid |
|---|---|
| Synonyms |
fmoc-l-pipecolic acid
MFCD00235898 2,2-Piperidinedicarboxylic acid, 2-(9H-fluoren-9-ylmethyl) ester (2S)-1-[(9H-Fluoren-9-ylmethoxy)carbonyl]-2-piperidinecarboxylic acid (2S)-1-[(9H-Fluoren-9-ylmethoxy)carbonyl]piperidine-2-carboxylic acid 2-[(9H-Fluoren-9-ylmethoxy)carbonyl]-2-piperidinecarboxylic acid Fmoc-β-Homo-Pro-OH (S)-1-FMOC-PIPERIDINE-2-CARBOXYLIC ACID Fmoc-HomoPro-OH N-Fmoc-L-pipecolic acid (S)-1,2-Piperidinedicarboxylic acid 1-(9H-fluoren-9-ylmethyl) ester Fmoc-HoPro-OH |
| Description | Fmoc-HoPro-OH is a proline derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.293±0.06 g/cm3 |
|---|---|
| Boiling Point | 553.7±50.0 °C at 760 mmHg |
| Melting Point | 161-162.5 ºC |
| Molecular Formula | C21H21NO4 |
| Molecular Weight | 351.396 |
| Flash Point | 288.7±30.1 °C |
| Exact Mass | 351.147064 |
| PSA | 66.84000 |
| LogP | 3.92 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.608 |
| Storage condition | 2-8°C |
| Water Solubility | Insuluble (8.8E-3 g/L) (25 ºC) |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi: Irritant; |
| RIDADR | UN 3077 9 / PGIII |
| WGK Germany | 3 |
| HS Code | 2933399090 |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |