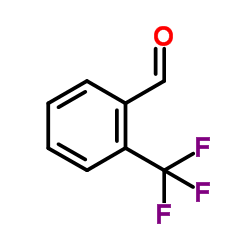
71145-03-4
| Name | 1,4-Dihydro-2,6-dimethyl-3-nitro-4-(2-trifluoromethylphenyl)-pyridine-5-carboxylic acid methyl ester |
|---|---|
| Synonyms |
(1,4-dihydro-2,6-dimethyl-5-nitro-4-[2-(trifluoromethyl)-phenyl]-3-pyridine carboxylic acid methyl ester
rac-Aziridin-2-carbonitril methyl-1,4-dihydro-2,6-dimethyl-5-nitro-4-(2-trifluoromethylphenyl)-pyridine-3-carboxylate (+/-)-Aziridin-2-carbonitril cyano-2 aziridine (-)-2,6-dimethyl-3-carbomethoxy-5-nitro-4-(2-trifluoromethylphenyl)-1,4-dihydropyridine 2-aziridinecarbonitrile MFCD00036697 2-cyano-2-aziridine (.+/-.)-Bay K 8644 nitrile of aziridine-2-carboxylic acid Bay K-8644 |
| Description | Bay K 8644, a dihydropyridine compound, is a specific L-type Ca2+ channel agonist. Bay K 8644 increases Ca2+ influx through sarcolemmal Ca2+ channels by increasing the open time of the channel[1]. |
|---|---|
| Related Catalog | |
| In Vitro | In newborn rat ventricular cardiomyocytes, Bay K 8644 (1 μM) treatment increases L-type calcium current density in 2-day-old cells. The higher increase of L-type calcium current density by Bay K 8644 in 2-day- than in 7-day-old cultured cells could be interpreted as the result of a difference in the phosphorylation level of calcium channels for each stage of development[2]. |
| In Vivo | A one time dose as low as 10 μg/kg of Bay K 8644 significantly elevates mean arterial pressure (MAP) in endotoxin-treated hypotensive rats while having minimal effects in normal rats. Bay K 8644 also causes a dose-dependent decrease in heart rate of 37% in endotoxin-treated rats and 39% in control rats[1]. |
| References |
| Density | 1.37g/cm3 |
|---|---|
| Boiling Point | 404.3ºC at 760 mmHg |
| Molecular Formula | C16H15F3N2O4 |
| Molecular Weight | 356.29700 |
| Flash Point | 198.3ºC |
| Exact Mass | 356.09800 |
| PSA | 84.15000 |
| LogP | 4.19940 |
| Index of Refraction | 1.545 |
| Storage condition | -20℃ |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319 |
| Precautionary Statements | P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi |
| Risk Phrases | 36/38 |
| Safety Phrases | 26-36 |
| RIDADR | NONH for all modes of transport |
|
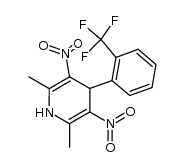
~37% 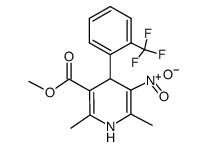
71145-03-4 |
| Literature: Bayer Aktiengesellschaft Patent: US4532248 A1, 1985 ; |
|
~33% 
71145-03-4 |
| Literature: Gorlitzer; Schmidt Archiv der Pharmazie, 1991 , vol. 324, # 10 p. 785 - 796 |
|
~21%
Detail
|
| Literature: Gorlitzer; Schmidt Archiv der Pharmazie, 1991 , vol. 324, # 10 p. 785 - 796 |
| Precursor 4 | |
|---|---|
| DownStream 0 | |