1947-37-1
| Name | tetragastrin |
|---|---|
| Synonyms |
Cholecystokinin Fragment 30-33 Amide
cholecystokinin-4 H-Trp-Met-Asp-Phe-NH2 L-Phenylalaninamide,L-tryptophyl-L-methionyl-L-a-aspartyl cholecystokinin tetrapeptide Cholecystokinin fragment 30-33 amide Cholecystokinin C-terminal tetrapeptide |
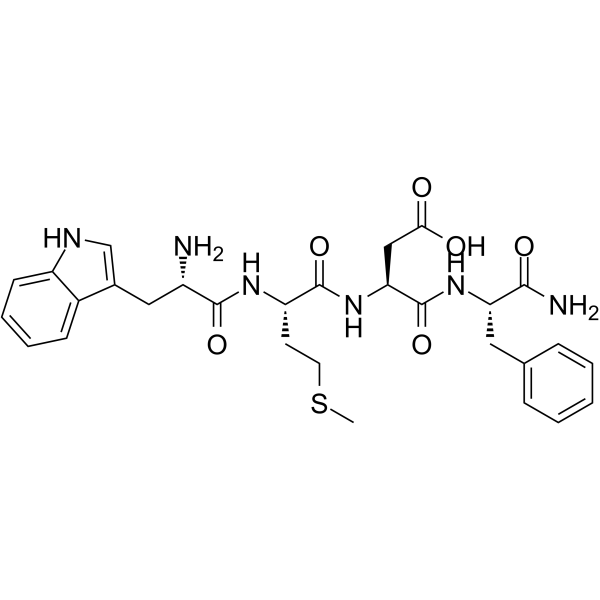
| Description | Tetragastrin (Cholecystokinin tetrapeptide; CCK-4) is the C-terminal tetrapeptide of gastrin. Tetragastrin can stimulate gastric secretion[1]. Tetragastrin is a Cholecystokinin (CCK-4) receptor agonist[2]. Gastric mucosal protection[3]. |
|---|---|
| Related Catalog | |
| In Vitro | The antagonist of histamine H2-receptors, Cimetidine inhibits the stimulatory effect of histamine in vitro and activates Tetragastrin stimulation of the adenylate cyclase activity. Tetragastrin and histamine activate adenylate cyclase of the rat gastric mucosa via different receptors[4]. |
| In Vivo | In inbred Wistar rats treated with N-methyl-N'-nitro-N-nitrosoguanidine, Tetragastrin (s.c.; 1 mg/kg; every other day) treatment significantly reduces the incidence and the number of adenocarcinomas, and has a significantly lower labelling index of the antral mucosa[1]. Tetragastrin has potential for enhancing gastric mucosal protection associated with mucus secretion and/or mucus synthesis on the surface mucosa of rat gastric mucosa. A significant increase in the mucin content was noted in the mucus gel and surface mucosal layer. An increase in mucin in the mucus gel and surface mucosa would thus appear due to the administration of Tetragastrin[3]. Animal Model: Seven-week-old male Wistar rats, each weighing approximately 160 g[3] Dosage: 12, 120, or 400 μg/kg Administration: Administered subcutaneously (s.c.); followed by 50% ethanol-induced gastric injury Result: Caused a significant increase in mucin content in the corpus mucosa and prevented 50% ethanol-induced gastric mucosal damage in a dose-dependent manner. |
| References |
| Density | 1.341g/cm3 |
|---|---|
| Boiling Point | 1159.2ºC at 760mmHg |
| Molecular Formula | C29H36N6O6S |
| Molecular Weight | 596.70 |
| Flash Point | 654.8ºC |
| Exact Mass | 730.27800 |
| PSA | 247.11000 |
| LogP | 4.67950 |
| Vapour Pressure | 0mmHg at 25°C |
| Index of Refraction | 1.643 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |