Efloxate
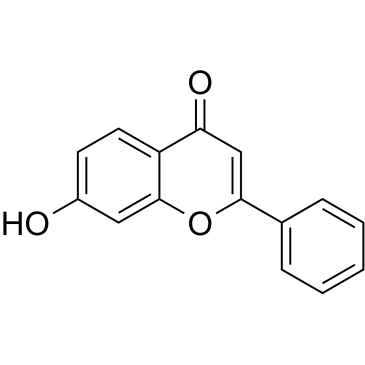
Efloxate structure
|
Common Name | Efloxate | ||
|---|---|---|---|---|
| CAS Number | 119-41-5 | Molecular Weight | 324.32700 | |
| Density | 1.265g/cm3 | Boiling Point | 486.5ºC at 760 mmHg | |
| Molecular Formula | C19H16O5 | Melting Point | 123-124° | |
| MSDS | N/A | Flash Point | 269.1ºC | |
Use of EfloxateEfloxate is a vasodilator, used to treat chronic coronary insufficiency and Angina pectoris, |
| Name | ethyl 2-(4-oxo-2-phenylchromen-7-yl)oxyacetate |
|---|---|
| Synonym | More Synonyms |
| Description | Efloxate is a vasodilator, used to treat chronic coronary insufficiency and Angina pectoris, |
|---|---|
| Related Catalog |
| Density | 1.265g/cm3 |
|---|---|
| Boiling Point | 486.5ºC at 760 mmHg |
| Melting Point | 123-124° |
| Molecular Formula | C19H16O5 |
| Molecular Weight | 324.32700 |
| Flash Point | 269.1ºC |
| Exact Mass | 324.10000 |
| PSA | 65.74000 |
| LogP | 3.40190 |
| Vapour Pressure | 1.28E-09mmHg at 25°C |
| Index of Refraction | 1.59 |
| Storage condition | 2-8℃ |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| HS Code | 2918990090 |
|---|
|
~% 
Efloxate CAS#:119-41-5 |
| Literature: Tobe, Masanori; Isobe, Yoshiaki; Goto, Yuso; Obara, Fumihiro; Tsuchiya, Masami; Matsui, Junko; Hirota, Kosaku; Hayashi, Hideya Bioorganic and Medicinal Chemistry, 2000 , vol. 8, # 8 p. 2037 - 2047 |
|
~% 
Efloxate CAS#:119-41-5 |
| Literature: Zwaagstra; Timmerman; Abdoelgafoer; Zhang European Journal of Medicinal Chemistry, 1996 , vol. 31, # 11 p. 861 - 874 |
|
~% 
Efloxate CAS#:119-41-5 |
| Literature: Da Re; Colleoni Annali di Chimica (Rome, Italy), 1959 , vol. 49, p. 1632,1636 |
| HS Code | 2918990090 |
|---|---|
| Summary | 2918990090. other carboxylic acids with additional oxygen function and their anhydrides, halides, peroxides and peroxyacids; their halogenated, sulphonated, nitrated or nitrosated derivatives. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Secondary assay to identify inhibitors of non-replicating M. tb using luciferase expr...
Source: Broad Institute
Target: N/A
External Id: 2157-04_Inhibitor_Dose_DryPowder_Activity_Set4
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Cytotoxicity counterscreen for inhibitors of SARS-CoV-2 cell entry
Source: NCGC
Target: N/A
External Id: TRND-SARS-CoV-2-cytotox-48hr
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Elucidation of physiology of non-replicating, drug-tolerant Mycobacterium tuberculosi...
Source: Broad Institute
Target: N/A
External Id: 2157-01_Inhibitor_Dose_DryPowder_Activity
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
| Corosanin |
| Efloxatum |
| Recordil |
| Oxyflavil |
| [(4-oxo-2-phenyl-4H-1-benzopyran-7-yl)oxy]-acetic acid,ethyl ester |
| (4-Oxo-2-phenyl-4H-chromen-7-yloxy)-essigsaeure-aethylester |
| Coril |
| EINECS 204-321-9 |
| Efloxate |
| (4-oxo-2-phenyl-4H-chromen-7-yloxy)-acetic acid ethyl ester |
| Angorlisin |
| Oxiflavil |
| Flacethyle |
| 7-ethoxycarbonylmethoxyflavone |
| Domucor |