(R)-3-Hydroxybutanoic acid sodium
Modify Date: 2025-08-21 14:06:50
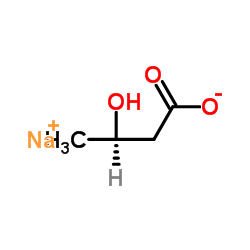
(R)-3-Hydroxybutanoic acid sodium structure
|
Common Name | (R)-3-Hydroxybutanoic acid sodium | ||
|---|---|---|---|---|
| CAS Number | 13613-65-5 | Molecular Weight | 126.086 | |
| Density | N/A | Boiling Point | 269.2ºCat 760 mmHg | |
| Molecular Formula | C4H7NaO3 | Melting Point | 149-155ºC(lit.) | |
| MSDS | USA | Flash Point | 121ºC | |
Use of (R)-3-Hydroxybutanoic acid sodium(R)-3-Hydroxybutanoic acid sodium ((R)-3-Hydroxybutyric acid) is a metabolite converted from acetoacetic acid catalyzed by 3-hydroxybutyrate dehydrogenase. (R)-3-Hydroxybutanoic acid sodium has applications as a nutrition source, and as a precursor for vitamins, antibiotics and pheromones[1][2]. |
| Name | Sodium (R)-3-hydroxybutanoate |
|---|---|
| Synonym | More Synonyms |
| Description | (R)-3-Hydroxybutanoic acid sodium ((R)-3-Hydroxybutyric acid) is a metabolite converted from acetoacetic acid catalyzed by 3-hydroxybutyrate dehydrogenase. (R)-3-Hydroxybutanoic acid sodium has applications as a nutrition source, and as a precursor for vitamins, antibiotics and pheromones[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | (R)-3-Hydroxybutanoic acid sodium is a metabolite converted from acetoacetic acid. Enhanced hepatic fatty acid oxidation results in the increased production of acetoacetic acid which is in turn converted to (R)-3-Hydroxybutanoic acid sodium by a reaction catalyzed by 3-hydroxybutyrate dehydrogenase[1]. |
| References |
| Boiling Point | 269.2ºCat 760 mmHg |
|---|---|
| Melting Point | 149-155ºC(lit.) |
| Molecular Formula | C4H7NaO3 |
| Molecular Weight | 126.086 |
| Flash Point | 121ºC |
| Exact Mass | 126.029289 |
| PSA | 60.36000 |
| Vapour Pressure | 0.000979mmHg at 25°C |
| InChIKey | NBPUSGBJDWCHKC-AENDTGMFSA-M |
| SMILES | CC(O)CC(=O)[O-].[Na+] |
| Storage condition | 2-8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Safety Phrases | 22-24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2918199090 |
| HS Code | 2918199090 |
|---|---|
| Summary | 2918199090 other carboxylic acids with alcohol function but without other oxygen function, their anhydrides, halides, peroxides, peroxyacids and their derivatives。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:6.5%。General tariff:30.0% |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: Absorbance-based primary biochemical high throughput screening assay to identify acti...
Source: The Scripps Research Institute Molecular Screening Center
Target: caspase-3 preproprotein [Homo sapiens]
External Id: PROCASPASE3_ACT_EPIABS_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule modulators of NR3A
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: SBCCG-A1015-NR3A-Primary-Assay
|
Total 101, Current Page 1 of 11
1
2
3
4
5
| (R)-(-)-3-Hydroxybutyric acid sodium salt |
| Sodium (3R)-3-hydroxybutanoate |
| sodium,(3R)-3-hydroxybutanoate |
| Butanoic acid, 3-hydroxy-, sodium salt, (3R)- (1:1) |
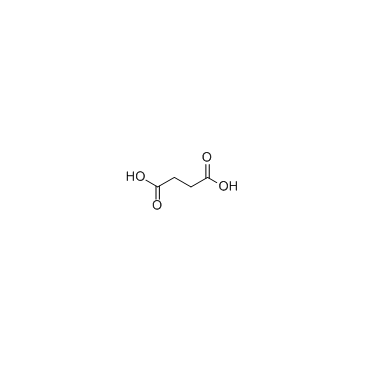 CAS#:110-15-6
CAS#:110-15-6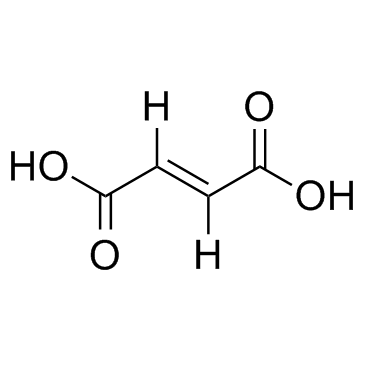 CAS#:110-17-8
CAS#:110-17-8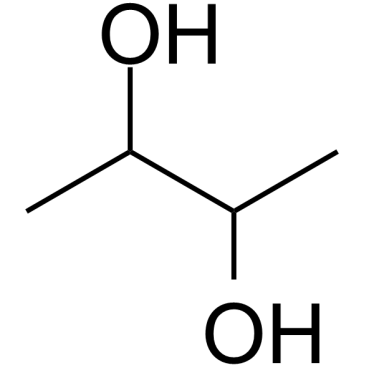 CAS#:513-85-9
CAS#:513-85-9 CAS#:625-72-9
CAS#:625-72-9
