Calanolide E
Modify Date: 2025-08-23 16:26:51
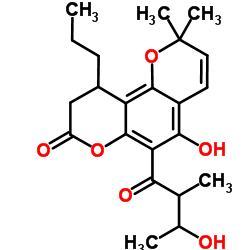
Calanolide E structure
|
Common Name | Calanolide E | ||
|---|---|---|---|---|
| CAS Number | 142566-61-8 | Molecular Weight | 388.45 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 537.1±50.0 °C at 760 mmHg | |
| Molecular Formula | C22H28O6 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 182.8±23.6 °C | |
Use of Calanolide ECalanolide E is a natural coumarin with weakly inhibitory towards the Bacilli strains with MIC values ranging from 0.25-0.50 mg/mL[1]. |
| Name | 5-Hydroxy-6-(3-hydroxy-2-methylbutanoyl)-2,2-dimethyl-10-propyl-9 ,10-dihydro-2H,8H-pyrano[2,3-f]chromen-8-one |
|---|---|
| Synonym | More Synonyms |
| Description | Calanolide E is a natural coumarin with weakly inhibitory towards the Bacilli strains with MIC values ranging from 0.25-0.50 mg/mL[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 537.1±50.0 °C at 760 mmHg |
| Molecular Formula | C22H28O6 |
| Molecular Weight | 388.45 |
| Flash Point | 182.8±23.6 °C |
| Exact Mass | 388.188599 |
| PSA | 93.06000 |
| LogP | 5.97 |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.553 |
| Hazard Codes | Xi |
|---|
| 5-Hydroxy-6-(3-hydroxy-2-methylbutanoyl)-2,2-dimethyl-10-propyl-9,10-dihydro-2H,8H-pyrano[2,3-f]chromen-8-one |
| 2H,8H-Benzo[1,2-b:3,4-b']dipyran-8-one, 9,10-dihydro-5-hydroxy-6-(3-hydroxy-2-methyl-1-oxobutyl)-2,2-dimethyl-10-propyl- |

