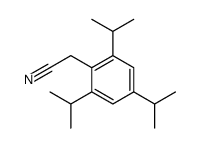
Avasimibe
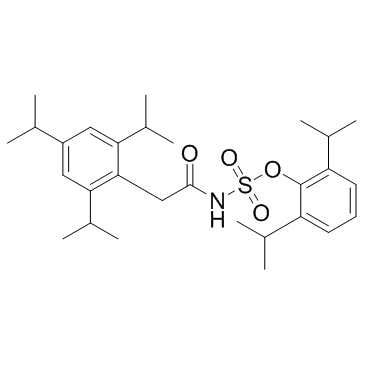
Avasimibe structure
|
Common Name | Avasimibe | ||
|---|---|---|---|---|
| CAS Number | 166518-60-1 | Molecular Weight | 501.721 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C29H43NO4S | Melting Point | 178-180° (Lee); mp 169.5-170.4° (Dozeman) | |
| MSDS | USA | Flash Point | N/A | |
Use of AvasimibeAvasimibe is an oral inhibitor of acyl-Coenzyme A:cholesterol acyltransferase (ACAT) with IC50s of 24 and 9.2 µM for ACAT1 and ACAT2, respectively. |
| Name | [[2,4,6-tris(1-methylethyl)phenyl]acetyl]-, 2,6-bis(1-methylethyl)phenyl ester] sulfamic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Avasimibe is an oral inhibitor of acyl-Coenzyme A:cholesterol acyltransferase (ACAT) with IC50s of 24 and 9.2 µM for ACAT1 and ACAT2, respectively. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Melting Point | 178-180° (Lee); mp 169.5-170.4° (Dozeman) |
| Molecular Formula | C29H43NO4S |
| Molecular Weight | 501.721 |
| Exact Mass | 501.291290 |
| PSA | 80.85000 |
| LogP | 9.34 |
| Appearance of Characters | white to tan |
| Index of Refraction | 1.529 |
| Storage condition | room temp |
| Water Solubility | DMSO: ≥40mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|---|
| HS Code | 2935009090 |
|
~77% 
Avasimibe CAS#:166518-60-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 39, # 26 p. 5031 - 5034 |
|
~% 
Avasimibe CAS#:166518-60-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 39, # 26 p. 5031 - 5034 |
|
~% 
Avasimibe CAS#:166518-60-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 39, # 26 p. 5031 - 5034 |
|
~% 
Avasimibe CAS#:166518-60-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 39, # 26 p. 5031 - 5034 |
|
~% 
Avasimibe CAS#:166518-60-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 39, # 26 p. 5031 - 5034 |
|
~% 
Avasimibe CAS#:166518-60-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 39, # 26 p. 5031 - 5034 |
| HS Code | 2935009090 |
|---|---|
| Summary | 2935009090 other sulphonamides VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:35.0% |
|
Statin therapy alone and in combination with an acyl-CoA:cholesterol O-acyltransferase inhibitor on experimental atherosclerosis.
Pathophysiol. Haemost. Thromb. 36(1) , 9-17, (2007) The ability to modify the enzymatic processes involved in promoting atherosclerotic plaque disruption and to serially monitor atherosclerotic evolution could provide novel information in the managemen... |
|
|
Coronary circulatory function in patients with the metabolic syndrome.
J. Nucl. Med. 52(9) , 1369-77, (2011) The metabolic syndrome affects 25% of the U.S. population and greatly increases the risk of diabetes and coronary artery disease (CAD). We tested the hypothesis that the metabolic syndrome is associat... |
|
|
Avasimibe and atorvastatin synergistically reduce cholesteryl ester content in THP-1 macrophages.
Eur. J. Pharmacol. 451(1) , 11-7, (2002) Evidence suggests that the inhibition of both acyl-CoA:cholesterol acyltransferase and hydroxymethyl glutaryl-CoA reductase causes a synergistic direct antiatherosclerotic effect on the vessel wall. T... |
| 2,6-Diisopropylphenyl [(2,4,6-triisopropylphenyl)acetyl]sulfamate |
| Sulfamic acid, N-[2-[2,4,6-tris(1-methylethyl)phenyl]acetyl]-, 2,6-bis(1-methylethyl)phenyl ester |
| [2,6-di(propan-2-yl)phenyl] N-[2-[2,4,6-tri(propan-2-yl)phenyl]acetyl]sulfamate |
| 2,6-di(propan-2-yl)phenyl {[2,4,6-tri(propan-2-yl)phenyl]acetyl}sulfamate |
| Avasimibe |
| CI-1011 |
![2-[2,4,6-tri(propan-2-yl)phenyl]acetyl chloride structure](https://image.chemsrc.com/caspic/050/52629-47-7.png)

![2-[2,4,6-tri(propan-2-yl)phenyl]acetic acid structure](https://image.chemsrc.com/caspic/441/4276-85-1.png)