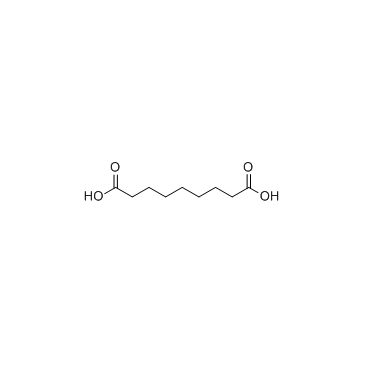
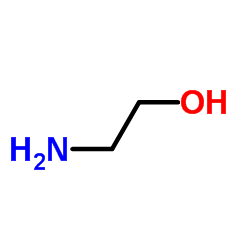
Adelmidrol
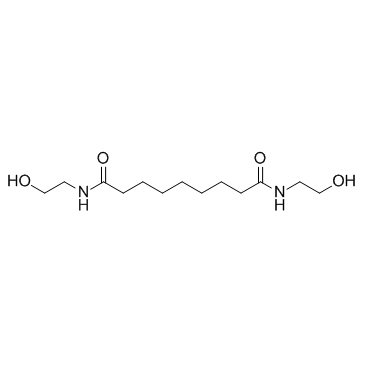
Adelmidrol structure
|
Common Name | Adelmidrol | ||
|---|---|---|---|---|
| CAS Number | 1675-66-7 | Molecular Weight | 274.35700 | |
| Density | 1.097g/cm3 | Boiling Point | 596.4ºC at 760mmHg | |
| Molecular Formula | C13H26N2O4 | Melting Point | 132-134 ºC | |
| MSDS | N/A | Flash Point | 314.5ºC | |
Use of AdelmidrolAdelmidrol exerts important anti-inflammatory effects that are partly dependent on PPARγ. Adelmidrol reduces NF-κB translocation, and COX-2 expression. |
| Name | N,N'-bis(2-hydroxyethyl)nonanediamide |
|---|---|
| Synonym | More Synonyms |
| Description | Adelmidrol exerts important anti-inflammatory effects that are partly dependent on PPARγ. Adelmidrol reduces NF-κB translocation, and COX-2 expression. |
|---|---|
| Related Catalog | |
| Target |
NF-κB COX-2 PPARγ |
| In Vitro | Adelmidrol is a palmitoylethanolamide analogue. Adelmidrol reduces NF-κB translocation, COX-2, and p-ERK expression; proinflammatory cytokine release; and the incidence of nitrotyrosine and poly(ADP)ribose in the colon[1]. |
| In Vivo | Adelmidrol (10 mg/kg, o.s.) reduces significantly the degree and severity of the macroscopic and histologic signs of colon injury. Moreover, 4 days after colitis induced by dinitrobenzene sulfonic acid (DNBS) treatment, all mice have diarrhea and a reduction in body weight (compared with the sham groups). Adelmidrol (10 mg/kg, o.s.) treatment significantly reduces the loss of body weight. The inflammatory bowel disease (IBD) induced by DNBS intrarectally administered is also characterized by an augmentation in myeloperoxidase (MPO) activity, an indicator of neutrophil accumulating in the colon. This is consistent with light microscopic observations showing the colon of vehicle-treated DNBS mice to contain a large number of neutrophils. In contrast, Adelmidrol (10 mg/kg, o.s.) significantly reduces the degree of polymorphonuclear cell infiltration (determined as reduction in MPO activity) in inflamed colon[1]. |
| Animal Admin | Mice[1] Male adult CD1 mice (25-30 g) and male mice (20-27 g) are placed in a controlled environment and maintained on a 12-hour light/dark cycle with food and water available ad libitum. Mice are casually divided into the following groups (10 in each group) (1)Sham+vehicle group: Vehicle solution (saline) is given by oral administration for 4 days. (2) Sham+Adelmidrol (10 mg/kg): Administered o.s. for 4 days. (3) DNBS+vehicle: Mice are injected by DNBS as described, and vehicle (saline) is given o.s. each day for 4 days, starting 60 minutes after the injection of DNBS. (4) DNBS+Adelmidrol (10 mg/kg): Mice are injected by DNBS as described, and Adelmidrol (10 mg/kg) is given o.s. each day, starting 60 minutes after administration of DNBS[1]. |
| References |
| Density | 1.097g/cm3 |
|---|---|
| Boiling Point | 596.4ºC at 760mmHg |
| Melting Point | 132-134 ºC |
| Molecular Formula | C13H26N2O4 |
| Molecular Weight | 274.35700 |
| Flash Point | 314.5ºC |
| Exact Mass | 274.18900 |
| PSA | 98.66000 |
| LogP | 0.71590 |
| Vapour Pressure | 1.04E-16mmHg at 25°C |
| Index of Refraction | 1.492 |
| InChIKey | PAHZPHDAJQIETD-UHFFFAOYSA-N |
| SMILES | O=C(CCCCCCCC(=O)NCCO)NCCO |
| Storage condition | 2-8℃ |
| Water Solubility | Soluble (60 g/L) (25 ºC) |
|
~77% 
Adelmidrol CAS#:1675-66-7 |
| Literature: LIFEGROUP S.p.A. Patent: EP550008 A2, 1993 ; |
|
~80% 
Adelmidrol CAS#:1675-66-7 |
| Literature: Lifegroup S.P.A. Patent: US5925678 A1, 1999 ; |
| Precursor 3 | |
|---|---|
| DownStream 0 | |
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: ULK1_INH_LUMI_1536_1X%INH PRUN
|
|
Name: Enzymatic assay of human HDAC6 with commercial peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808149
|
|
Name: Enzymatic assay of human HDAC6 with custom peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808150
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of VER...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4513082
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of Cac...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303805
|
|
Name: Phenotypic Assay to Identify Small Molecules that Upregulate Production of hCFTR in H...
Source: Southern Research Institute
Target: CFTR
External Id: CF Folding
|
|
Name: SARS-CoV-2 3CL-Pro protease inhibition percentage at 20µM by FRET kind of response f...
Source: ChEMBL
Target: Replicase polyprotein 1ab
External Id: CHEMBL4495582
|
| Adelmidrol |
| UNII-1BUC3685QU |