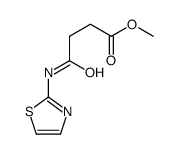
Butanoicacid, 4-oxo-4-(2-thiazolylamino)
Modify Date: 2025-08-25 11:23:48
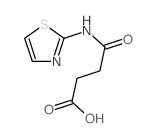
Butanoicacid, 4-oxo-4-(2-thiazolylamino) structure
|
Common Name | Butanoicacid, 4-oxo-4-(2-thiazolylamino) | ||
|---|---|---|---|---|
| CAS Number | 19692-00-3 | Molecular Weight | 200.21500 | |
| Density | 1.507g/cm3 | Boiling Point | 417.4ºC at 760mmHg | |
| Molecular Formula | C7H8N2O3S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 206.3ºC | |
| Name | 4-oxo-4-(1,3-thiazol-2-ylamino)butanoic acid |
|---|---|
| Synonym | More Synonyms |
| Density | 1.507g/cm3 |
|---|---|
| Boiling Point | 417.4ºC at 760mmHg |
| Molecular Formula | C7H8N2O3S |
| Molecular Weight | 200.21500 |
| Flash Point | 206.3ºC |
| Exact Mass | 200.02600 |
| PSA | 107.53000 |
| LogP | 1.01940 |
| Vapour Pressure | 3.88E-08mmHg at 25°C |
| Index of Refraction | 1.639 |
| InChIKey | SVMGZMBKMJRZLI-UHFFFAOYSA-N |
| SMILES | O=C(O)CCC(=O)Nc1nccs1 |
| HS Code | 2934100090 |
|---|
|
~88% 
Butanoicacid, 4... CAS#:19692-00-3 |
| Literature: Chu, Guo-Hua; Gu, Minghua; Cassel, Joel A.; Belanger, Serge; Graczyk, Thomas M.; DeHaven, Robert N.; Conway-James, Nathalie; Koblish, Michael; Little, Patrick J.; DeHaven-Hudkins, Diane L.; Dolle, Roland E. Bioorganic and Medicinal Chemistry Letters, 2007 , vol. 17, # 7 p. 1951 - 1955 |
|
~91% 
Butanoicacid, 4... CAS#:19692-00-3 |
| Literature: Burdulene; Stumbryavichyute; Talaikite; Vladyko; Boreko; Korobchenko Pharmaceutical Chemistry Journal, 1996 , vol. 30, # 11 p. 680 - 682 |
| Precursor 3 | |
|---|---|
| DownStream 0 | |
| HS Code | 2934100090 |
|---|---|
| Summary | 2934100090 other compounds containing an unfused thiazole ring (whether or not hydrogenated) in the structure VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:20.0% |
|
Name: Cell survival assay for modulators of telomere damage signalling
Source: 15378
Target: N/A
External Id: TELO_02
|
|
Name: Discovery of Small Molecules to Inhibit Human Cytomegalovirus Nuclear Egress
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: HCMV UL50
External Id: HMS1262
|
|
Name: Screen for inhibitors of RMI FANCM (MM2) intereaction
Source: 11908
Target: N/A
External Id: RMI-FANCM-MM2
|
|
Name: Inhibitors of CDC25B-CDK2/CyclinA interaction
Source: Center for Chemical Genomics, University of Michigan
External Id: MScreen:TargetID_600
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
|
Name: A screen for compounds that inhibit viral RNA polymerase binding and polymerization a...
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: Chain A, Poliovirus Polymerase With Gtp
External Id: HMS750
|
Total 9, Current Page 1 of 1
1
| N-Thiazol-2-yl-succinamidsaeure |
| N-thiazol-2-yl-succinamic acid |