5-Bromo-1-methylisatin
Modify Date: 2025-08-23 11:25:34
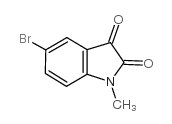
5-Bromo-1-methylisatin structure
|
Common Name | 5-Bromo-1-methylisatin | ||
|---|---|---|---|---|
| CAS Number | 2058-72-2 | Molecular Weight | 240.05300 | |
| Density | 1.729 g/cm3 | Boiling Point | 362.6ºC at 760 mmHg | |
| Molecular Formula | C9H6BrNO2 | Melting Point | 171 °C | |
| MSDS | N/A | Flash Point | 173.1ºC | |
| Name | 5-bromo-1-methylindole-2,3-dione |
|---|---|
| Synonym | More Synonyms |
| Density | 1.729 g/cm3 |
|---|---|
| Boiling Point | 362.6ºC at 760 mmHg |
| Melting Point | 171 °C |
| Molecular Formula | C9H6BrNO2 |
| Molecular Weight | 240.05300 |
| Flash Point | 173.1ºC |
| Exact Mass | 238.95800 |
| PSA | 37.38000 |
| LogP | 1.67320 |
| Vapour Pressure | 1.91E-05mmHg at 25°C |
| Index of Refraction | 1.64 |
| InChIKey | GEEDYJPPYNIZLX-UHFFFAOYSA-N |
| SMILES | CN1C(=O)C(=O)c2cc(Br)ccc21 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| HS Code | 2933990090 |
|---|
| Precursor 9 | |
|---|---|
| DownStream 1 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: qHTS screening for TAG (triacylglycerol) accumulators in algae
Source: 11812
Target: N/A
External Id: FATTTLab-Algae-Lipid
|
|
Name: Fluorescence-based counterscreen assay for HCV NS3 helicase inhibitors of a ChemBridg...
Source: 1102
Target: N/A
External Id: 20130627FPNS3INTERFERECB
|
|
Name: Cell survival assay for modulators of telomere damage signalling
Source: 15378
Target: N/A
External Id: TELO_02
|
|
Name: Small-molecule inhibitors of ST2 (IL1RL1)
Source: 20881
Target: interleukin-1 receptor-like 1 isoform [homo sapiens]
External Id: ST2_IL33_Inhibitors_Primary_Screening_77700
|
|
Name: Fluorescence polarization based primary biochemical high throughput screening assay o...
Source: 1102
Target: NS3 [Hepatitis C virus]
External Id: 20130624FPNS3DACB
|
|
Name: Alphascreen assay for small molecules abrogating mHTT-CaM Interaction
Source: 24983
Target: Huntingtin
External Id: KUHTS-Muma KU-CaM-Htt INH-01
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
Total 9, Current Page 1 of 1
1
| 1-N-methyl-5-bromoisatin |
| 5-bromo-1-methyl-1H-indole-2,3-dione |
| INDOLE-2,3-DIONE,5-BROMO-1-METHYL |
| 1-methyl-5-bromoindoline-2,3-dione |
| 5-bromo-1-methylindoline-2,3-dione |
| 5-bromo-1-methyl-2,3-dihydro-1H-indole-2,3-dione |
| 5-bromo-1-methyl-indole-2,3-dione |
| 5-Bromo-1-methyl-isatin |
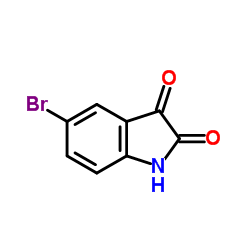 CAS#:87-48-9
CAS#:87-48-9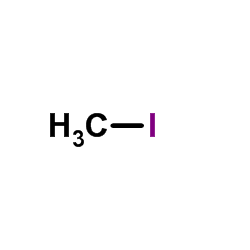 CAS#:74-88-4
CAS#:74-88-4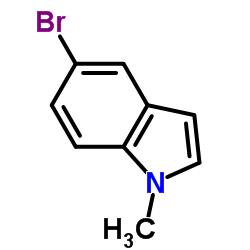 CAS#:10075-52-2
CAS#:10075-52-2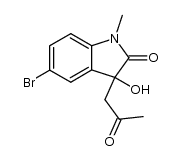 CAS#:107864-80-2
CAS#:107864-80-2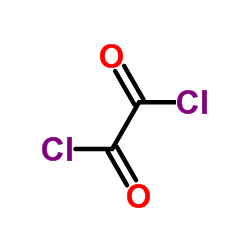 CAS#:79-37-8
CAS#:79-37-8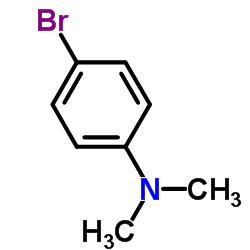 CAS#:586-77-6
CAS#:586-77-6 CAS#:2058-74-4
CAS#:2058-74-4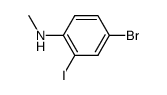 CAS#:869486-64-6
CAS#:869486-64-6 CAS#:35452-54-1
CAS#:35452-54-1![8-bromo-5-methyl-2H-[1,2,4]triazino[5,6-b]indole-3-thione structure](https://image.chemsrc.com/caspic/452/23563-33-9.png) CAS#:23563-33-9
CAS#:23563-33-9
