Thiobenzamide
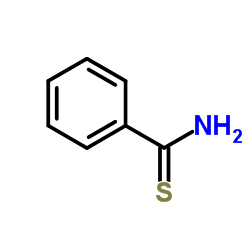
Thiobenzamide structure
|
Common Name | Thiobenzamide | ||
|---|---|---|---|---|
| CAS Number | 2227-79-4 | Molecular Weight | 137.202 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 245.0±23.0 °C at 760 mmHg | |
| Molecular Formula | C7H7NS | Melting Point | 113-117 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 102.0±22.6 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
| Name | benzenecarbothioamide |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 245.0±23.0 °C at 760 mmHg |
| Melting Point | 113-117 °C(lit.) |
| Molecular Formula | C7H7NS |
| Molecular Weight | 137.202 |
| Flash Point | 102.0±22.6 °C |
| Exact Mass | 137.029922 |
| PSA | 58.11000 |
| LogP | 1.49 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.653 |
| InChIKey | QIOZLISABUUKJY-UHFFFAOYSA-N |
| SMILES | NC(=S)c1ccccc1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R25 |
| Safety Phrases | S45 |
| RIDADR | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | CV5860000 |
| Packaging Group | III |
| Hazard Class | 6.1 |
| HS Code | 2930909090 |
| HS Code | 2930909090 |
|---|---|
| Summary | 2930909090. other organo-sulphur compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Cytotoxic activity of 3-(5-phenyl-3H-[1,2,4]dithiazol-3-yl)chromen-4-ones and 4-oxo-4H-chromene-3-carbothioic acid N-phenylamides.
Eur. J. Med. Chem. 45 , 790-4, (2010) 6/6,7-Substituted-3-formylchromones (8a-g) were reacted with 2 equivalents thiobenzamide (9) in refluxing toluene to furnish substituted-3-(5-phenyl-3H-[1,2,4]dithiazol-3-yl)chromen-4-ones (10a-g) in ... |
|
|
Potent inhibition of alcohol self-administration in alcohol-preferring rats by a κ-opioid receptor antagonist.
J. Pharmacol. Exp. Ther. 350(1) , 171-80, (2014) A substituted aryl amide derivative of 6-naltrexamine--17-cyclopropylmethyl-3,14β-dihydroxy-4,5α-epoxy-6β-[(4'-trimethylfluoro)benzamido]morphinan-hydrochloride--(compound 5), previously shown to be a... |
|
|
Facile synthesis of nitriles via manganese oxide promoted oxidative dehydrosulfurization of primary thioamides.
Chem. Commun. (Camb.) 48(91) , 11247-9, (2012) In the presence of manganese oxides, dehydrosulfurization of various kinds of primary thioamides including aromatic, heterocyclic, and aliphatic ones efficiently proceeded to give the corresponding ni... |
| Benzenecarbothioamide |
| EINECS 218-765-6 |
| phenylthioamide |
| MFCD00008060 |
| Benzamide,thio |
| Benzenecarbimidothioic acid |
| aminophenylmethane-1-thione |
| Thiobenzamide |
| Benzenecarboximidothioic acid |
| Benzothioamide |
| Benzothiamide |

