Kavain
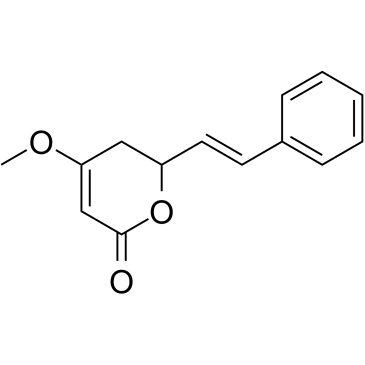
Kavain structure
|
Common Name | Kavain | ||
|---|---|---|---|---|
| CAS Number | 3155-48-4 | Molecular Weight | 230.259 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 432.6±45.0 °C at 760 mmHg | |
| Molecular Formula | C14H14O3 | Melting Point | 142-148ºC | |
| MSDS | Chinese USA | Flash Point | 184.6±23.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of KavainKavain is a class of kavalactone isolated from Piper methysticum, which has anxiolytic and sedative properties in animals and humans. Kavain positively modulated γ-Aminobutyric acid type A (GABAA) receptor[1]. |
| Name | Kavain |
|---|---|
| Synonym | More Synonyms |
| Description | Kavain is a class of kavalactone isolated from Piper methysticum, which has anxiolytic and sedative properties in animals and humans. Kavain positively modulated γ-Aminobutyric acid type A (GABAA) receptor[1]. |
|---|---|
| In Vitro | Two-electrode voltage clamp technique is used to characterize the functional properties of the major anxiolytic kavalactone, Kavain at human recombinant α1β2, β2γ2L, αxβ2γ2L, α1βxγ2L and α4β2δ γ-Aminobutyric acid type A receptors (GABAARs) expressed in Xenopus oocytes. Kavain positively modulates all receptors regardless of the subunit composition, but the degree of enhancement is greater at α4β2δ than at α1β2γ2L GABAARs. The modulatory effect of Kkavain is unaffected by flumazenil, indicating that Kavain does not enhance GABAARs via the classical benzodiazepine binding site. The β3N265M point mutation which has been previously shown to profoundly decrease anaesthetic sensitivity, also diminishes Kavain-mediated potentiation[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 432.6±45.0 °C at 760 mmHg |
| Melting Point | 142-148ºC |
| Molecular Formula | C14H14O3 |
| Molecular Weight | 230.259 |
| Flash Point | 184.6±23.3 °C |
| Exact Mass | 230.094299 |
| PSA | 35.53000 |
| LogP | 1.69 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.565 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
|
Fatal kavalactone intoxication by suicidal intravenous injection.
Forensic Sci. Int. 249 , e7-11, (2015) Kavalactones are a group of compounds found in kava, a beverage or extract prepared from the rhizome of the kava plant (Piper methysticum). Traditionally kava extracts have been used for their anxioly... |
|
|
Kavalactones fail to inhibit alcohol dehydrogenase in vitro.
Phytomedicine 13(3) , 192-5, (2006) In recent years, Kava kava (Piper methysticum, Forst. f., Piperaceae), a folkloric beverage and popular herbal remedy, has been implicated in a number of liver failure cases. Many hypotheses as to the... |
|
|
Kavalactones Yangonin and Methysticin induce apoptosis in human hepatocytes (HepG2) in vitro.
Phytother Res. 25(3) , 417-23, (2011) While cases of severe kava hepatotoxicity have been reported, studies examining the toxicity of individual kavalactones are limited. The present study examined the in vitro hepatotoxicity of kavain, m... |
| DL-Kawain |
| 2H-Pyran-2-one, 5,6-dihydro-4-methoxy-6-[(E)-2-phenylethenyl]- |
| 4-Methoxy-6-[(E)-2-phenylvinyl]-5,6-dihydro-2H-pyran-2-one |
| trans-5,6-Dihydro-4-methoxy-6-(2-phenylethenyl)-2H-pyran-2-one,DL-Kawain |
| KAVAIN,DL |
| UNII:5L1NI60TGB |
| Kawain,DL |
| Kawain |
| Kavain |
| 2H-Pyran-2-one, 5,6-dihydro-4-methoxy-6-(2-phenylethenyl)-, (E)-(±)- |
| MFCD00270446 |
| Kawain, DL- |
| 2H-Pyran-2-one, 5,6-dihydro-4-methoxy-6-styryl- |
| Kavain, dl- |
| DL-KAVAIN |