m-Oxalotoluidide
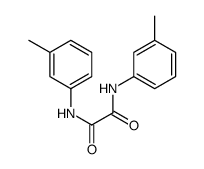
m-Oxalotoluidide structure
|
Common Name | m-Oxalotoluidide | ||
|---|---|---|---|---|
| CAS Number | 3551-75-5 | Molecular Weight | 268.31000 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C16H16N2O2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
| Name | N,N'-bis(3-methylphenyl)oxamide |
|---|---|
| Synonym | More Synonyms |
| Molecular Formula | C16H16N2O2 |
|---|---|
| Molecular Weight | 268.31000 |
| Exact Mass | 268.12100 |
| PSA | 58.20000 |
| LogP | 3.02660 |
| InChIKey | SGPZWRDIDBJXET-UHFFFAOYSA-N |
| SMILES | Cc1cccc(NC(=O)C(=O)Nc2cccc(C)c2)c1 |
| HS Code | 2924299090 |
|---|
|
~23% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Habash, Maha; Taha, Mutasem O. Bioorganic and Medicinal Chemistry, 2011 , vol. 19, # 16 p. 4746 - 4771 |
|
~58% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Kubicova, Lenka; Waisser, Karel; Kunes, Jiri; Kralova, Katarina; Odlerova, Zelmira; Slosarek, Milan; Janota, Jiri; Svoboda, Zbynek Molecules, 2000 , vol. 5, # 5 p. 714 - 726 |
|
~% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Oef.Sv., , # 10 p. 15 |
|
~% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Synthetic Communications, , vol. 22, # 7 p. 1081 - 1085 |
|
~% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Journal of Organic Chemistry USSR (English Translation), , vol. 1, p. 1223 - 1225 Zhurnal Organicheskoi Khimii, , vol. 1, p. 1212 - 1214 |
|
~% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Recueil des Travaux Chimiques des Pays-Bas, , vol. 61, p. 647,658 |
|
~% 
m-Oxalotoluidide CAS#:3551-75-5 |
| Literature: Chemische Berichte, , vol. 40, p. 2653 |
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Name: Inhibition of rabbit muscle glycogen phosphorylase 1a assessed as release of phosphat...
Source: ChEMBL
Target: Glycogen phosphorylase, muscle form
External Id: CHEMBL1817502
|
|
Name: Inhibition of rabbit muscle glycogen phosphorylase 1a assessed as release of phosphat...
Source: ChEMBL
Target: Glycogen phosphorylase, muscle form
External Id: CHEMBL1817501
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
| m-Oxalotoluidide |
| Oxalsaeure-di-m-toluidid |
| N.N'-Di-m-tolyl-oxamid |
| N,N'-di-m-tolyloxalamide |
| N,N'-Di-m-tolyl-oxalamid |

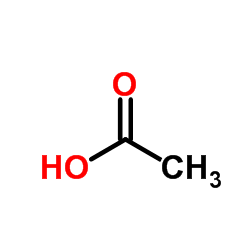


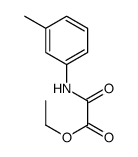
 CAS#:7030-60-6
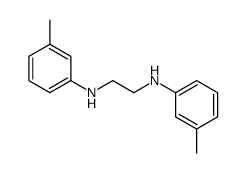
CAS#:7030-60-6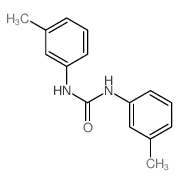 CAS#:620-50-8
CAS#:620-50-8 CAS#:401585-34-0
CAS#:401585-34-0