isatidine
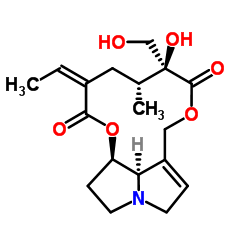
isatidine structure
|
Common Name | isatidine | ||
|---|---|---|---|---|
| CAS Number | 480-54-6 | Molecular Weight | 351.394 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 583.2±50.0 °C at 760 mmHg | |
| Molecular Formula | C18H25NO6 | Melting Point | 208-211ºC(lit.) | |
| MSDS | Chinese USA | Flash Point | 306.5±30.1 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of isatidineRetrorsine is a naturally occurring toxic pyrrolizidine alkaloid. Retrorsine can bind with DNA and inhibits the proliferative capacity of hepatocytes[1][2]. |
| Name | retrorsine |
|---|---|
| Synonym | More Synonyms |
| Description | Retrorsine is a naturally occurring toxic pyrrolizidine alkaloid. Retrorsine can bind with DNA and inhibits the proliferative capacity of hepatocytes[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Retrorsine (60-240 μM; 24 hours) significantly reduces HSEC-CYP3A4 cells viability, depletes GSH, and increases formation of pyrrole-protein adducts[3]. Cell Viability Assay[3] Cell Line: HSEC-CYP3A4 cells Concentration: 60 μM, 120 μM , 240 μM Incubation Time: 24 hours Result: Significantly decreased cell viability. |
| In Vivo | Retrorsine (30 mg/kg; i.p.; twice) impairs liver regeneration in the PBL model not only by an S or G2/M phase block, but also by a block located before the G1/S transition of the cell cycle[1]. Animal Model: Male Wistar rats (180±20 g), portal branch ligation (PBL) model[1] Dosage: 30 mg/kg Administration: Intraperitoneal injection, twice, separated by 2-week interval Result: Strongly impaired the liver weight gain, protein and DNA synthesis as well as induction of cell cycle related proteins in the regenerating lobes after PBL. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 583.2±50.0 °C at 760 mmHg |
| Melting Point | 208-211ºC(lit.) |
| Molecular Formula | C18H25NO6 |
| Molecular Weight | 351.394 |
| Flash Point | 306.5±30.1 °C |
| Exact Mass | 351.168182 |
| PSA | 96.30000 |
| LogP | -0.14 |
| Vapour Pressure | 0.0±3.7 mmHg at 25°C |
| Index of Refraction | 1.590 |
| InChIKey | BCJMNZRQJAVDLD-CQRYIUNCSA-N |
| SMILES | CC=C1CC(C)C(O)(CO)C(=O)OCC2=CCN3CCC(OC1=O)C23 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H300 |
| Precautionary Statements | P264-P301 + P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T |
| Risk Phrases | 25 |
| Safety Phrases | S22;S45;S36/S37/S39 |
| RIDADR | UN 1544 |
| RTECS | VH7525000 |
| Packaging Group | II |
| Hazard Class | 6.1(a) |
| Precursor 1 | |
|---|---|
| DownStream 1 | |
|
Diversity of pyrrolizidine alkaloids in native and invasive Senecio pterophorus (Asteraceae): implications for toxicity.
Phytochemistry 108 , 137-46, (2014) Changes in plant chemical defenses after invasion could have consequences on the invaded ecosystems by modifying the interactions between plants and herbivores and facilitating invasion success. Howev... |
|
|
Involvement of organic cation transporter 1 and CYP3A4 in retrorsine-induced toxicity.
Toxicology 322 , 34-42, (2014) Retrorsine (RTS) is a hepatotoxic pyrrolizidine alkaloid present in plants of the Senecio genus. The present study is aimed at clarifying the role of organic cation transporters (OCTs) in the liver di... |
|
|
Are effects of common ragwort in the Ames test caused by pyrrolizidine alkaloids?
Mutat. Res. 778 , 1-10, (2015) It has previously been demonstrated by others that acetone extracts of Senecio jacobaea (syn. Jacobaea vulgaris, common or tansy ragwort) test positive in the Salmonella/microsome mutagenicity test (A... |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: USP8 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 8
External Id: USP8 FAST DUB HTS Primary
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: USP17 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 17 like family member 5
External Id: USP17 FAST DUB HTS Primary
|
|
Name: USP7 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 7
External Id: USP7 FAST DUB HTS Primary
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
| (15Z)-12,18-Dihydroxysenecionan-11,16-dione |
| isatidine |
| trans-15-Ethylidene-12b-hydroxy-12a-hydroxymethyl-13b-methylsenec-1-enine |
| b-Longilobine |
| 12,18-dihydroxy-senecionane-11,16-dione |
| 12,18-Dihydroxysenecionan-11,16-dione |
| β-Longilobine |
| cis-Retronecic acid ester of retronecine |
| Retrorsin |
| (3Z,5R,6S,14aR,14bR)-3-Ethylidene-6-hydroxy-6-(hydroxymethyl)-5-methyl-3,4,5,6,9,11,13,14,14a,14b-decahydro[1,6]dioxacyclododecino[2,3,4-gh]pyrrolizine-2,7-dione |
| MFCD00013331 |
 CAS#:69102-07-4
CAS#:69102-07-4 CAS#:74164-84-4
CAS#:74164-84-4