5-hydroxyuracil
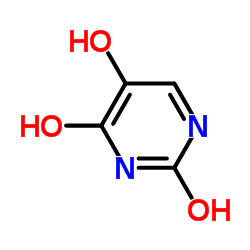
5-hydroxyuracil structure
|
Common Name | 5-hydroxyuracil | ||
|---|---|---|---|---|
| CAS Number | 496-76-4 | Molecular Weight | 128.086 | |
| Density | 1.8±0.1 g/cm3 | Boiling Point | 534.5±53.0 °C at 760 mmHg | |
| Molecular Formula | C4H4N2O3 | Melting Point | 300ºC | |
| MSDS | Chinese USA | Flash Point | 277.0±30.9 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 5-hydroxyuracil |
|---|---|
| Synonym | More Synonyms |
| Density | 1.8±0.1 g/cm3 |
|---|---|
| Boiling Point | 534.5±53.0 °C at 760 mmHg |
| Melting Point | 300ºC |
| Molecular Formula | C4H4N2O3 |
| Molecular Weight | 128.086 |
| Flash Point | 277.0±30.9 °C |
| Exact Mass | 128.022186 |
| PSA | 85.95000 |
| LogP | -2.10 |
| Appearance of Characters | crystalline | light yellow |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.719 |
| Water Solubility | Soluble in DMSO (Sparingly), Methanol (Sparingly). |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
|
Hydrolytic pathway of 5-fluorouracil in aqueous solutions for clinical use.
J. Pharm. Biomed. Anal. 98 , 446-62, (2014) The purpose of the study was to investigate the degradation pathway of 5-fluorouracil (FU) in the situation of commercial formulations for clinical use, namely FU dissolved in sodium hydroxide (NaOH) ... |
|
|
Mutational analysis of the damage-recognition and catalytic mechanism of human SMUG1 DNA glycosylase.
Nucleic Acids Res. 32(17) , 5291-302, (2004) Single-strand selective monofunctional uracil-DNA glycosylase (SMUG1), previously thought to be a backup enzyme for uracil-DNA glycosylase, has recently been shown to excise 5-hydroxyuracil (hoU), 5-h... |
|
|
Base-promoted reaction of 5-hydroxyuracil derivatives with peroxyl radicals.
Org. Lett. 12(18) , 4130-3, (2010) Addition of millimolar amounts of a weak base (pyridines) dramatically accelerates the reaction with peroxyl radicals of two biologically relevant uracil derivatives, 5-hydroxyuracil (HU) and 5-hydrox... |
| Pyrimidinetriol |
| 2,4,6-ThihydroxypyriMidine |
| pyrimidine-2,4,5-triol |
| Isobarbitursaeure |
| 2,4,5-pyrimidinetriol |
| Isobarbituric Acid |
| 2,4,5-Trihydroxypyrimidine,5-Hydroxyuracil,Isobarbituric acid |
| 5-hydroxyuracil |
| 2,4,5-Trihydroxypyrimidine |
| Fluorouracil impurity B |
| 5-hydroxyisouracil |
| 5-Hydroxypyrimidine-2,4(1H,3H)-dione |
| 5-Hydroxy-2,4(1H,3H)-pyrimidinedione |
| TRIFLUOROACETIC ACID REAGENTPLUS |
| EINECS 207-829-9 |
| MFCD00082987 |
| Isobarbituric acid (VAN) (8CI) |