Thidiazuron
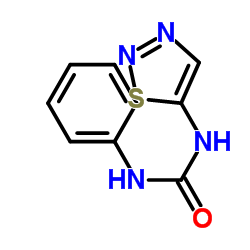
Thidiazuron structure
|
Common Name | Thidiazuron | ||
|---|---|---|---|---|
| CAS Number | 51707-55-2 | Molecular Weight | 220.25 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 410.5±55.0 °C at 760 mmHg | |
| Molecular Formula | C9H8N4OS | Melting Point | 213°C | |
| MSDS | Chinese USA | Flash Point | 202.1±31.5 °C | |
| Symbol |


GHS07, GHS09 |
Signal Word | Warning | |
Use of ThidiazuronThidiazuron is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | thidiazuron |
|---|---|
| Synonym | More Synonyms |
| Description | Thidiazuron is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog | |
| In Vitro | Thidiazuron 是一种植物生长调节剂,用作 Murashige 和 Skoog 培养基等培养基的补充剂,用于微繁殖。Thidiazuron 促进植物器官发生(芽再生)和植物再生。 |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 410.5±55.0 °C at 760 mmHg |
| Melting Point | 213°C |
| Molecular Formula | C9H8N4OS |
| Molecular Weight | 220.25 |
| Flash Point | 202.1±31.5 °C |
| Exact Mass | 220.041885 |
| PSA | 95.15000 |
| LogP | 1.81 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.722 |
| InChIKey | HFCYZXMHUIHAQI-UHFFFAOYSA-N |
| SMILES | O=C(Nc1ccccc1)Nc1cnns1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |


GHS07, GHS09 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335-H410 |
| Precautionary Statements | P273-P305 + P351 + P338-P391-P501 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S22-S26-S36 |
| RIDADR | UN 3077 9 / PGIII |
| WGK Germany | 3 |
| RTECS | YU1395000 |
|
Mining biologically-active molecules for inhibitors of fatty acid amide hydrolase (FAAH): Identification of phenmedipham and amperozide as FAAH inhibitors
Bioorg. Med. Chem. Lett. 19 , 6793-6, (2009) The screening of known medicinal agents against new biological targets has been shown to be a valuable approach for revealing new pharmacology of marketed compounds. Recently, carbamate, urea and keto... |
|
|
Role of TDZ in the quick regeneration of multiple shoots from nodal explant of Vitex trifolia L.--an important medicinal plant.
Appl. Biochem. Biotechnol. 168(5) , 957-66, (2012) The effect of thidiazuron (TDZ) has been investigated in shoot multiplication for a simple, efficient, rapid, and commercially applicable regeneration protocol of an important medicinal plant, Vitex t... |
|
|
Changes of gentiopicroside synthesis during somatic embryogenesis in Gentiana macrophylla.
Planta Med. 75 , 1618-1624, (2009) IN VITRO plant regeneration of Gentiana macrophylla Pall. and determination of gentiopicroside content during somatic embryogenesis are described in the present work. The highest percentage of embryog... |
| 1-Phenyl-3-(1,2,3-thiadiazol-5-yl)urea |
| 1-phenyl-3-(thiadiazol-5-yl)urea |
| Methanol, 1-(phenylimino)-1-(1,2,3-thiadiazol-5-ylamino)-, (E)- |
| Thidiazuron |
| Urea, N-phenyl-N'-1,2,3-thiadiazol-5-yl- |
| N-phenyl-N’-1,2,3-thiadiazol-5-ylurea |
| MFCD00078723 |
| EINECS 257-356-7 |
| N'-Phenyl-N-1,2,3-thiadiazol-5-ylcarbamimidic acid |

