Lobaric acid
Modify Date: 2025-09-18 14:26:03
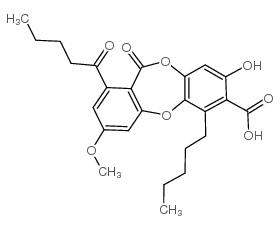
Lobaric acid structure
|
Common Name | Lobaric acid | ||
|---|---|---|---|---|
| CAS Number | 522-53-2 | Molecular Weight | 456.48500 | |
| Density | 1.265g/cm3 | Boiling Point | 667.9ºC at 760mmHg | |
| Molecular Formula | C25H28O8 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 225.7ºC | |
Use of Lobaric acidLobaric acid is a depsidone metabolite that has been isolated from Stereocaulon lichen species with antioxidant, antiproliferative, antiviral, and enzyme inhibitory activites. It scavenges superoxide radicals in a cell-free assay (IC50 = 97.9 μmol) and inhibits proliferation in a panel of leukemia, colorectal, gastric, breast, ovarian, prostate, pancreatic, and lung cancer cell lines (EC50s = 15.2-63.9 μg/ml). Lobaric acid inhibits protein tyrosine phosphatase 1B (PTP1B; IC50 = 0.87 μM for the human recombinant enzyme) and production of 12(S)-HETE (Item No. 34570) by 12(S)-lipoxygenase (IC50 = 28.5 μM). In vivo, lobaric acid (250 μM) decreases lesion number, but not lesion diameter, in tobacco leaves infected with tobacco mosaic virus (TMV). |
| Name | 3-hydroxy-9-methoxy-6-oxo-7-pentanoyl-1-pentylbenzo[b][1,4]benzodioxepine-2-carboxylic acid |
|---|---|
| Synonym | More Synonyms |
| Density | 1.265g/cm3 |
|---|---|
| Boiling Point | 667.9ºC at 760mmHg |
| Molecular Formula | C25H28O8 |
| Molecular Weight | 456.48500 |
| Flash Point | 225.7ºC |
| Exact Mass | 456.17800 |
| PSA | 119.36000 |
| LogP | 5.52950 |
| Index of Refraction | 1.579 |
| InChIKey | JHEWMLHQNRHTQX-UHFFFAOYSA-N |
| SMILES | CCCCCc1c2c(cc(O)c1C(=O)O)OC(=O)c1c(cc(OC)cc1C(=O)CCCC)O2 |
| Precursor 0 | |
|---|---|
| DownStream 2 | |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Inhibition of PTP1B (1 to 298 residues) (unknown origin) using p-nitrophenylphosphate...
Source: ChEMBL
Target: Tyrosine-protein phosphatase non-receptor type 1
External Id: CHEMBL4150189
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Confirmed inhibitors of the Choline Transporter (CHT)
Source: 1043
Target: high affinity choline transporter 1 [Homo sapiens]
External Id: SAID_488997
|
Total 157, Current Page 1 of 16
1
2
3
4
5
| Usnetic acid |
| Lobaric acid |
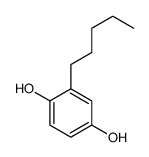 CAS#:4693-32-7
CAS#:4693-32-7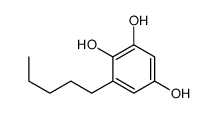 CAS#:89244-04-2
CAS#:89244-04-2