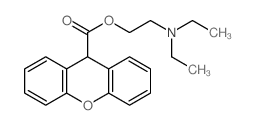
Methantheline Bromide
Modify Date: 2025-08-23 06:49:16
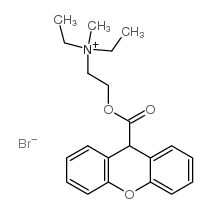
Methantheline Bromide structure
|
Common Name | Methantheline Bromide | ||
|---|---|---|---|---|
| CAS Number | 53-46-3 | Molecular Weight | 420.34000 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C21H26BrNO3 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of Methantheline BromideMethantheline Bromide is a synthetic antispasmodic. It is used to relieve cramps or spasms of the stomach, intestines, and bladder. Methantheline is used to treat intestine or stomach ulcers (peptic ulcer disease), intestine problems, pancreatitis, gastritis, biliary dyskinesia, pylorosplasm, or urinary problems. |
| Name | methantheline bromide (200 mg) |
|---|---|
| Synonym | More Synonyms |
| Molecular Formula | C21H26BrNO3 |
|---|---|
| Molecular Weight | 420.34000 |
| Exact Mass | 419.11000 |
| PSA | 35.53000 |
| LogP | 0.95780 |
| InChIKey | PQMWYJDJHJQZDE-UHFFFAOYSA-M |
| SMILES | CC[N+](C)(CC)CCOC(=O)C1c2ccccc2Oc2ccccc21.[Br-] |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|---|
| HS Code | 2932999099 |
|
~% 
Methantheline B... CAS#:53-46-3 |
| Literature: US2659732 , ; DE948333 , ; |
|
~% 
Methantheline B... CAS#:53-46-3 |
| Literature: Pharmaceutical Bulletin, , vol. 3, p. 417,420 |
| Precursor 3 | |
|---|---|
| DownStream 0 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: p53 small molecule agonists, cell-based qHTS assay: qHTS cell viability counter scree...
Source: 824
Target: N/A
External Id: P53600
|
|
Name: p53 small molecule agonists, cell-based qHTS assay with rat liver microsomes: qHTS ce...
Source: 824
Target: N/A
External Id: P53MS958
|
|
Name: p53 small molecule agonists, cell-based qHTS assay with rat liver microsomes: Summary
Source: 824
External Id: P53MS482
|
Total 374, Current Page 1 of 38
1
2
3
4
5
| asabaine |
| Methanthelinium bromide |
| mtb51 |
| diethyl-methyl-[2-(xanthene-9-carbonyloxy)-ethyl]-ammonium,bromide |
| sc2910 |
| ulcine |
| N,N-diethyl-N-methyl-2-[(9H-xanthen-9-ylcarbonyl)oxy]-ethanaminium |
| methanthelinium bromide |
| banthin |
| Diaethyl-methyl-[2-(xanthen-9-carbonyloxy)-aethyl]-ammonium,Bromid |
| avagal |
| Ulcine Bromide |
| methantheline bromide |
| doladene |
| metaxan |
| banthine |
| vagamin |