Irigenin
Modify Date: 2025-08-20 18:14:46
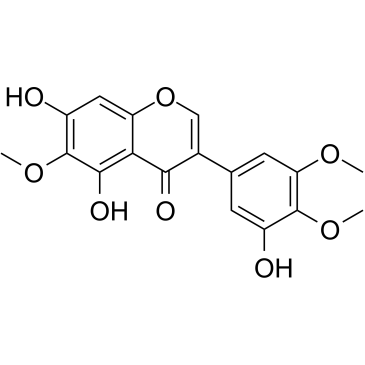
Irigenin structure
|
Common Name | Irigenin | ||
|---|---|---|---|---|
| CAS Number | 548-76-5 | Molecular Weight | 360.31500 | |
| Density | 1.461g/cm3 | Boiling Point | 646.1ºC at 760mmHg | |
| Molecular Formula | C18H16O8 | Melting Point | 189-192ºC | |
| MSDS | N/A | Flash Point | 237.7ºC | |
Use of IrigeninIrigenin is a is a lead compound, and mediates its anti-metastatic effect by specifically and selectively blocking α9β1 and α4β1 integrins binding sites on C-C loop of Extra Domain A (EDA). Irigenin shows anti-cancer properties. It sensitizes TRAIL-induced apoptosis via enhancing pro-apoptotic molecules in gastric cancer cells[1]. |
| Name | irigenin |
|---|---|
| Synonym | More Synonyms |
| Description | Irigenin is a is a lead compound, and mediates its anti-metastatic effect by specifically and selectively blocking α9β1 and α4β1 integrins binding sites on C-C loop of Extra Domain A (EDA). Irigenin shows anti-cancer properties. It sensitizes TRAIL-induced apoptosis via enhancing pro-apoptotic molecules in gastric cancer cells[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Irigenin specifically targets α9β1 and α4β1 integrin binding sites on Extra Domain A (EDA) comprising LEU46, PHE47, PRO48, GLU58, LEU59 and GLN60 in its C-C loop. Irigenin binds to the C-C loop of EDA, thereby blocking its interaction with integrins on the cell surface and thus abrogating subsequent Epithelial-Mesenchymal transition[1]. |
| References |
| Density | 1.461g/cm3 |
|---|---|
| Boiling Point | 646.1ºC at 760mmHg |
| Melting Point | 189-192ºC |
| Molecular Formula | C18H16O8 |
| Molecular Weight | 360.31500 |
| Flash Point | 237.7ºC |
| Exact Mass | 360.08500 |
| PSA | 118.59000 |
| LogP | 2.60260 |
| Index of Refraction | 1.649 |
| InChIKey | TUGWPJJTQNLKCL-UHFFFAOYSA-N |
| SMILES | COc1cc(-c2coc3cc(O)c(OC)c(O)c3c2=O)cc(O)c1OC |
| Storage condition | 2-8C |
| HS Code | 2914509090 |
|---|
| Precursor 0 | |
|---|---|
| DownStream 2 | |
| HS Code | 2914509090 |
|---|---|
| Summary | HS:2914509090 other ketones with other oxygen function VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:5.5% General tariff:30.0% |
|
Name: Estrogenic activity in human T47D cells assessed as drug level causing stimulation of...
Source: ChEMBL
Target: T47D
External Id: CHEMBL977512
|
|
Name: Estrogenic activity in human MCF7 cells assessed as drug level causing stimulation of...
Source: ChEMBL
Target: MCF7
External Id: CHEMBL977511
|
|
Name: Estrogenic activity in human T47D cells assessed as drug level causing stimulation of...
Source: ChEMBL
Target: T47D
External Id: CHEMBL977513
|
|
Name: Estrogenic activity in human MCF7 cells assessed as drug level causing stimulation of...
Source: ChEMBL
Target: MCF7
External Id: CHEMBL977510
|
|
Name: Primary high throughput screening by co-culture imaging for identification hits as a ...
Source: 23209
Target: N/A
External Id: UIHTS20180925
|
|
Name: Inhibition of cow milk xanthine oxidase at 50 ug/mL
Source: ChEMBL
Target: Xanthine dehydrogenase/oxidase
External Id: CHEMBL1015630
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
|
Name: The chemical genetic matrix (CGM) dataset as reported in Wildenhain et al. (2015) Pre...
Source: 11924
Target: N/A
External Id: CGM data for Cell Systems paper Dec 2015
|
Total 10, Current Page 1 of 1
1
| 5,7-dihydroxy-3-(3-hydroxy-4,5-dimethoxyphenyl)-6-methoxychromen-4-one |
| 7,5,3'-Trihydroxy-6,4',5'-trimethoxyisoflavon |
| 5,7,3'-trihydroxy-6,4',5'-trimethoxyisoflavone |
| Irigenin |
| EINECS 208-958-3 |
 CAS#:64-18-6
CAS#:64-18-6 CAS#:487-71-8
CAS#:487-71-8
