Roseoside
Modify Date: 2025-08-21 16:35:56
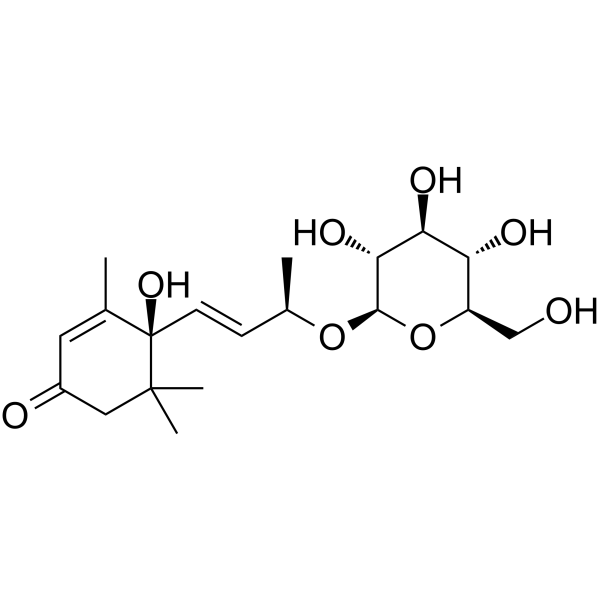
Roseoside structure
|
Common Name | Roseoside | ||
|---|---|---|---|---|
| CAS Number | 54835-70-0 | Molecular Weight | 386.437 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 595.9±50.0 °C at 760 mmHg | |
| Molecular Formula | C19H30O8 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 207.8±23.6 °C | |
Use of RoseosideRoseoside is a compound isolated from the leaves of Elaeocarpus japonicus[1]. |
| Name | (2R,3E)-4-[(1S)-1-Hydroxy-2,6,6-trimethyl-4-oxo-2-cyclohexen-1-yl ]-3-buten-2-yl β-D-glucopyranoside |
|---|---|
| Synonym | More Synonyms |
| Description | Roseoside is a compound isolated from the leaves of Elaeocarpus japonicus[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 595.9±50.0 °C at 760 mmHg |
| Molecular Formula | C19H30O8 |
| Molecular Weight | 386.437 |
| Flash Point | 207.8±23.6 °C |
| Exact Mass | 386.194061 |
| PSA | 136.68000 |
| LogP | -0.98 |
| Vapour Pressure | 0.0±3.8 mmHg at 25°C |
| Index of Refraction | 1.580 |
| InChIKey | SWYRVCGNMNAFEK-SUHXYMONSA-N |
| SMILES | CC1=CC(=O)CC(C)(C)C1(O)C=CC(C)OC1OC(CO)C(O)C(O)C1O |
| Hazard Codes | Xi |
|---|
| Precursor 0 | |
|---|---|
| DownStream 1 | |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: Absorbance-based primary biochemical high throughput screening assay to identify acti...
Source: The Scripps Research Institute Molecular Screening Center
Target: caspase-3 preproprotein [Homo sapiens]
External Id: PROCASPASE3_ACT_EPIABS_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule modulators of NR3A
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: SBCCG-A1015-NR3A-Primary-Assay
|
Total 104, Current Page 1 of 11
1
2
3
4
5
| Blumenol A b-D-glucopyranoside |
| roseoside A |
| Roseoside I |
| roseoside II |
| (2R,3E)-4-[(1S)-1-Hydroxy-2,6,6-trimethyl-4-oxo-2-cyclohexen-1-yl]-3-buten-2-yl β-D-glucopyranoside |
 CAS#:189351-15-3
CAS#:189351-15-3
