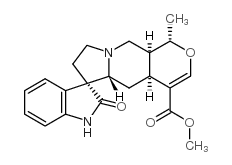
Pteropodine
Modify Date: 2024-01-16 15:46:46

Pteropodine structure
|
Common Name | Pteropodine | ||
|---|---|---|---|---|
| CAS Number | 5629-60-7 | Molecular Weight | 368.43 | |
| Density | 1.33g/cm3 | Boiling Point | 555.2ºC at 760mmHg | |
| Molecular Formula | C21H24N2O4 | Melting Point | 217-219ºC | |
| MSDS | N/A | Flash Point | 289.6ºC | |
Use of PteropodinePteropodine (compound 3) is a monoterpenoid oxindole alkaloid that can be isolated from Hamelia patens. Pteropodine modulates the functions of M1 muscarinic and 5-HT2 receptors. Pteropodine has antioxidant and antimutagenic properties[1]. |
| Name | Uncarine C |
|---|---|
| Synonym | More Synonyms |
| Description | Pteropodine (compound 3) is a monoterpenoid oxindole alkaloid that can be isolated from Hamelia patens. Pteropodine modulates the functions of M1 muscarinic and 5-HT2 receptors. Pteropodine has antioxidant and antimutagenic properties[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.33g/cm3 |
|---|---|
| Boiling Point | 555.2ºC at 760mmHg |
| Melting Point | 217-219ºC |
| Molecular Formula | C21H24N2O4 |
| Molecular Weight | 368.43 |
| Flash Point | 289.6ºC |
| Exact Mass | 368.17400 |
| PSA | 67.87000 |
| LogP | 2.13840 |
| Index of Refraction | 1.635 |
| Precursor 0 | |
|---|---|
| DownStream 1 | |
| Isospeciophylline |
| PTEROPODINE |
| 2'-keto-1-methyl-spiro[1,4a,5,5a,7,8,10,10a-octahydropyrano[3,4-f]indolizine-6,3'-indoline]-4-carboxylic acid methyl ester |
| methyl 1-methyl-2'-oxo-spiro[1,4a,5,5a,7,8,10,10a-octahydropyrano[3,4-f]indolizine-6,3'-1H-indole]-4-carboxylate |
 CAS#:4697-68-1
CAS#:4697-68-1