N-Hydroxybenzenecarboximidamide
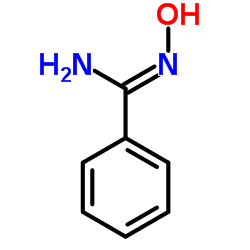
N-Hydroxybenzenecarboximidamide structure
|
Common Name | N-Hydroxybenzenecarboximidamide | ||
|---|---|---|---|---|
| CAS Number | 613-92-3 | Molecular Weight | 136.151 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 307.4±25.0 °C at 760 mmHg | |
| Molecular Formula | C7H8N2O | Melting Point | 77°C | |
| MSDS | Chinese USA | Flash Point | 139.7±23.2 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
| Name | benzamidoxime |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 307.4±25.0 °C at 760 mmHg |
| Melting Point | 77°C |
| Molecular Formula | C7H8N2O |
| Molecular Weight | 136.151 |
| Flash Point | 139.7±23.2 °C |
| Exact Mass | 136.063660 |
| PSA | 58.61000 |
| LogP | 1.15 |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.574 |
| InChIKey | MXOQNVMDKHLYCZ-UHFFFAOYSA-N |
| SMILES | NC(=NO)c1ccccc1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATAMUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H315-H319-H335 |
| Precautionary Statements | P261-P301 + P310-P305 + P351 + P338 |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R20/21/22;R36/37/38 |
| Safety Phrases | S22-S36/37/39 |
| RIDADR | 2811 |
| RTECS | CY6920000 |
| HS Code | 2925290090 |
| HS Code | 2925290090 |
|---|---|
| Summary | 2925290090 other imines and their derivatives; salts thereof。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:6.5%。General tariff:30.0% |
|
A novel and specific fluorescence reaction for uracil.
Anal. Chim. Acta 674(2) , 234-8, (2010) Facile and specific methods to quantify a nucleobase in biological samples are of great importance for diagnosing disorders in nucleic acid metabolism. In the present study, a novel fluorogenic reacti... |
|
|
Characteristics of the microsomal N-hydroxylation of benzamidine to benzamidoxime.
Xenobiotica 17(6) , 659-67, (1987) 1. A simple and fast h.p.l.c. analysis of benzamidoxime formed by microsomal N-hydroxylation of benzamidine is presented which is well suited for the determination of the N-oxygenation activity of mic... |
|
|
Biotransformation of benzamidine and benzamidoxime in vivo.
Arch. Pharm. (Weinheim) 326(10) , 807-12, (1993) After administration of benzamidine (1) or benzamidoxime (2), respectively, to rats and rabbits, plasma from rats and rabbits as well as urine from rats were examined for the presence of benzamidoxime... |
| EINECS 210-361-8 |
| BENZAMIDE OXIME |
| benzohydroxamamide |
| Benzamidoxim |
| MFCD06656049 |
| N-Hydroxybenzenecarboximidamide |
| N'-hydroxybenzimdamide |
| benzenylaminoxime |
| benzamidoxide |
| N'-hydroxy-benzene-carboximidamide |
| BENZAMIDOXIME |
| N-Hydroxybenzamidine |
| Benzamidoxine |
| benzenylamidoxime |
| BENZAMIDOXIME HYDROCHLORIDE |

