Allomatrine
Modify Date: 2025-08-23 16:08:01
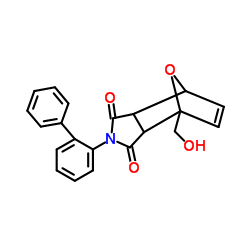
Allomatrine structure
|
Common Name | Allomatrine | ||
|---|---|---|---|---|
| CAS Number | 641-39-4 | Molecular Weight | 347.364 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 638.7±55.0 °C at 760 mmHg | |
| Molecular Formula | C15H24N2O | Melting Point | 106-107 ℃ | |
| MSDS | N/A | Flash Point | 340.1±31.5 °C | |
Use of AllomatrineAllomatrine ((+)-Allomatrine) is an alkaloid from the bark of Sophora japonica. Allomatrine has antinociceptive properties mediated mainly through the activation ofκ-opioid receptors[1][2]. |
| Name | 4-(2-Biphenylyl)-1-(hydroxymethyl)-10-oxa-4-azatricyclo[5.2.1.02,6]dec-8-ene-3,5-dione |
|---|---|
| Synonym | More Synonyms |
| Description | Allomatrine ((+)-Allomatrine) is an alkaloid from the bark of Sophora japonica. Allomatrine has antinociceptive properties mediated mainly through the activation ofκ-opioid receptors[1][2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 638.7±55.0 °C at 760 mmHg |
| Melting Point | 106-107 ℃ |
| Molecular Formula | C15H24N2O |
| Molecular Weight | 347.364 |
| Flash Point | 340.1±31.5 °C |
| Exact Mass | 347.115753 |
| PSA | 66.84000 |
| LogP | 2.09 |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.666 |
| InChIKey | ZSBXGIUJOOQZMP-KYEXWDHISA-N |
| SMILES | O=C1CCCC2C3CCCN4CCCC(CN12)C34 |
| Storage condition | 2-8C |
| Precursor 4 | |
|---|---|
| DownStream 0 | |
Total 8, Current Page 1 of 1
1
| 13S-HYDROXY-6Z,9Z,11E-OCTADECATRIENOIC ACID |
| 4-(2-Biphenylyl)-1-(hydroxymethyl)-10-oxa-4-azatricyclo[5.2.1.0]dec-8-ene-3,5-dione |
| 13-hydroxy-(6Z,9Z,11E,13S)-6,9,11-octadecatrienoic acid |
| 13(S)-HYDROXY-9(Z),11(E),15(Z)-OCTADECATRIENOIC ACID |
| (13S,6Z,9Z,11E)-hydroxyoctadeca-6,9,11-trienoic acid |
| Allomatrine |
| 13-hydroxy-6,9,11-octadecatrienoic acid |
| (+)-Allomatrin |
| 13(S)-HOT |
| 13(S)-HYDROXYOCTADECA-6Z,9Z,11E-TRIENOIC ACID |
| 13(S)-HOTE |
| (6Z,9Z,11E)-13(S)-hydroxyoctadeca-6,9,11-trienoic acid |
 CAS#:519-02-8
CAS#:519-02-8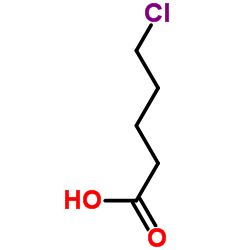 CAS#:1119-46-6
CAS#:1119-46-6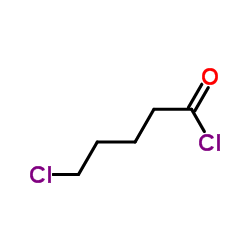 CAS#:1575-61-7
CAS#:1575-61-7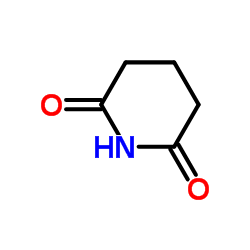 CAS#:1121-89-7
CAS#:1121-89-7
