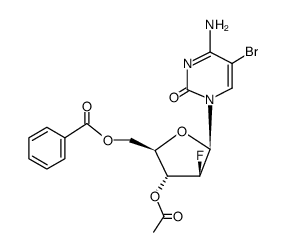
2'-fluoro-5-bromo-1-beta-D-arabinofuranosylcytosine
Modify Date: 2025-08-29 21:51:09
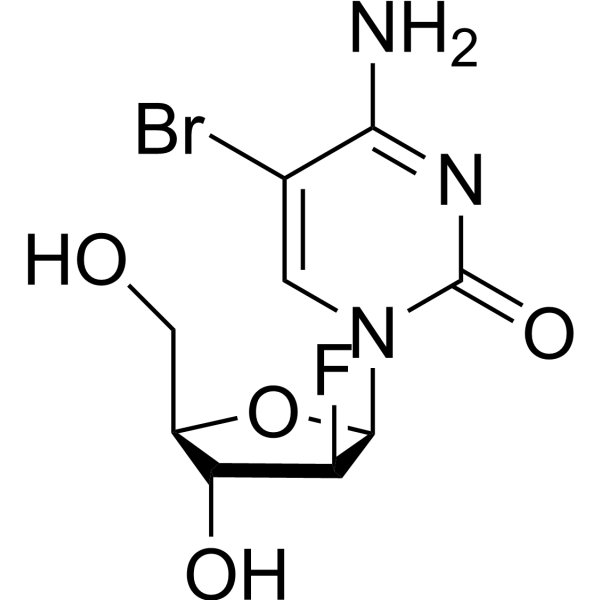
2'-fluoro-5-bromo-1-beta-D-arabinofuranosylcytosine structure
|
Common Name | 2'-fluoro-5-bromo-1-beta-D-arabinofuranosylcytosine | ||
|---|---|---|---|---|
| CAS Number | 69123-93-9 | Molecular Weight | 324.10 | |
| Density | 2.21 g/cm3 | Boiling Point | 513.3ºC at 760 mmHg | |
| Molecular Formula | C9H11BrFN3O4 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 264.3ºC | |
Use of 2'-fluoro-5-bromo-1-beta-D-arabinofuranosylcytosine5-Bromo-2’-deoxy-2’-fluoro-beta-D-arabinocytidine is a cytidine nucleoside analog. Cytidine analogs have a mechanism of inhibiting DNA methyltransferases (such as Zebularine, HY-13420), and have potential anti-metabolic and anti-tumor activities[1]. |
| Name | 4-amino-5-bromo-1-[(2R,3S,4R,5R)-3-fluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidin-2-one |
|---|---|
| Synonym | More Synonyms |
| Description | 5-Bromo-2’-deoxy-2’-fluoro-beta-D-arabinocytidine is a cytidine nucleoside analog. Cytidine analogs have a mechanism of inhibiting DNA methyltransferases (such as Zebularine, HY-13420), and have potential anti-metabolic and anti-tumor activities[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 2.21 g/cm3 |
|---|---|
| Boiling Point | 513.3ºC at 760 mmHg |
| Molecular Formula | C9H11BrFN3O4 |
| Molecular Weight | 324.10 |
| Flash Point | 264.3ºC |
| Exact Mass | 338.98700 |
| PSA | 130.83000 |
| Index of Refraction | 1.741 |
| InChIKey | DPTQLSGNVUCAMZ-BYPJNBLXSA-N |
| SMILES | Nc1nc(=O)n(C2OC(CO)C(O)C2F)cc1Br |
|
~% 
2'-fluoro-5-bro... CAS#:69123-93-9 |
| Literature: Sloan Kettering Institute for Cancer Research Patent: US4211773 A1, 1980 ; Title/Abstract Full Text Show Details Sloan-Kettering Institute for Cancer Research Patent: US4594339 A1, 1986 ; |
| Precursor 1 | |
|---|---|
| DownStream 0 | |
Total 33, Current Page 1 of 4
1
2
3
4
| 2'-Fluoro-5-bromo-aracytosine |