Schisanhenol
Modify Date: 2024-01-02 17:12:38
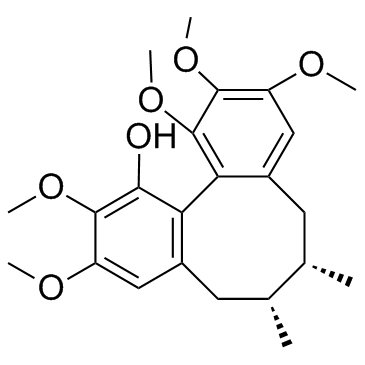
Schisanhenol structure
|
Common Name | Schisanhenol | ||
|---|---|---|---|---|
| CAS Number | 69363-14-0 | Molecular Weight | 402.481 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 563.9±50.0 °C at 760 mmHg | |
| Molecular Formula | C23H30O6 | Melting Point | 130 °C | |
| MSDS | N/A | Flash Point | 294.8±30.1 °C | |
Use of SchisanhenolSchisanhenol is a natural compound solated from Schisandra rubriflora; UGT2B7 UDP-glucuronosyltransferases inhibitor.IC50 value:Target: in vitro: Schisanhenol exhibited strong inhibition toward UGT2B7, with the residual activity to be 7.9% of control activity [1]. The BAECs were cultured with ox-LDL (200 microg/ml) in the presence and absence of Sal (10 and 50 micromol L(- 1)) for 24 h. The cytotoxicity of ox-LDL was evaluated by LDH leakage, cell viability and morphological change. Cell apoptosis was estimated by DNA ladder, chromatin condensation, and flow cytometry assay. The intracellular ROS production was detected by using DCF, a ROS probe, with laser confocal microscopy and flow cytometry. Sal was shown to reduce LDH leakage and increase cell viability. Sal also attenuated ox-LDL-induced BAECs apoptosis as indicated in typical internucleosomal DNA degradation (DNA ladder), condensed chromatin, and the sub-G1 peak appearance in flow cytometry assay [2].in vivo: Sal significantly impeded production of MDA and loss of ATPase activity induced by reoxygenation following anoxia. Oral administration of Sal induced increase of cytosol glutathione-peroxidase of brain in mice under the condition of reoxygenation following anoxia [4]. |
| Name | Schisanhenol |
|---|---|
| Synonym | More Synonyms |
| Description | Schisanhenol is a natural compound solated from Schisandra rubriflora; UGT2B7 UDP-glucuronosyltransferases inhibitor.IC50 value:Target: in vitro: Schisanhenol exhibited strong inhibition toward UGT2B7, with the residual activity to be 7.9% of control activity [1]. The BAECs were cultured with ox-LDL (200 microg/ml) in the presence and absence of Sal (10 and 50 micromol L(- 1)) for 24 h. The cytotoxicity of ox-LDL was evaluated by LDH leakage, cell viability and morphological change. Cell apoptosis was estimated by DNA ladder, chromatin condensation, and flow cytometry assay. The intracellular ROS production was detected by using DCF, a ROS probe, with laser confocal microscopy and flow cytometry. Sal was shown to reduce LDH leakage and increase cell viability. Sal also attenuated ox-LDL-induced BAECs apoptosis as indicated in typical internucleosomal DNA degradation (DNA ladder), condensed chromatin, and the sub-G1 peak appearance in flow cytometry assay [2].in vivo: Sal significantly impeded production of MDA and loss of ATPase activity induced by reoxygenation following anoxia. Oral administration of Sal induced increase of cytosol glutathione-peroxidase of brain in mice under the condition of reoxygenation following anoxia [4]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 563.9±50.0 °C at 760 mmHg |
| Melting Point | 130 °C |
| Molecular Formula | C23H30O6 |
| Molecular Weight | 402.481 |
| Flash Point | 294.8±30.1 °C |
| Exact Mass | 402.204254 |
| PSA | 66.38000 |
| LogP | 5.34 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.536 |
| HS Code | 2942000000 |
|---|
|
~% 
Schisanhenol CAS#:69363-14-0 |
| Literature: Chemical & Pharmaceutical Bulletin, , vol. 28, # 8 p. 2422 - 2427 |
| Precursor 1 | |
|---|---|
| DownStream 0 | |
| HS Code | 2942000000 |
|---|
| (+)-gomisin k3 |
| Dibenzo[a,c]cycloocten-1-ol, 5,6,7,8-tetrahydro-2,3,10,11,12-pentamethoxy-6,7-dimethyl-, (6S,7R)- |
| HMS2214M09 |
| (6S,7R)-2,3,10,11,12-Pentamethoxy-6,7-dimethyl-5,6,7,8-tetrahydrodibenzo[a,c][8]annulen-1-ol |
| UNII:80Y5907NIW |