2-Cyanobenzaldehyde
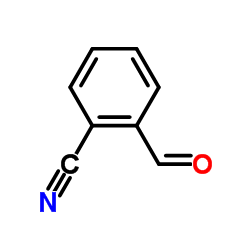
2-Cyanobenzaldehyde structure
|
Common Name | 2-Cyanobenzaldehyde | ||
|---|---|---|---|---|
| CAS Number | 7468-67-9 | Molecular Weight | 131.131 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 284.9±23.0 °C at 760 mmHg | |
| Molecular Formula | C8H5NO | Melting Point | 103-105 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 126.1±22.6 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 2-Cyanobenzaldehyde |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 284.9±23.0 °C at 760 mmHg |
| Melting Point | 103-105 °C(lit.) |
| Molecular Formula | C8H5NO |
| Molecular Weight | 131.131 |
| Flash Point | 126.1±22.6 °C |
| Exact Mass | 131.037109 |
| PSA | 40.86000 |
| LogP | 1.45 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.552 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H312-H315-H319-H332-H335 |
| Precautionary Statements | P261-P280-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R20/21/22;R36/37/38 |
| Safety Phrases | S26-S37/39-S36/37/39 |
| RIDADR | UN 3439 6.1/PG 3 |
| WGK Germany | 3 |
| Packaging Group | III |
| Hazard Class | 6.1 |
| HS Code | 2926909090 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2926909090 |
|---|---|
| Summary | HS:2926909090 other nitrile-function compounds VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:30.0% |
|
Diastereoselective one-pot tandem synthesis of 3-substituted isoindolinones: a mechanistic investigation.
J. Org. Chem. 75(17) , 5882-7, (2010) The mechanism of a base-catalyzed one-pot reaction of 2-cyanobenzaldehyde and primary nitroalkanes, to produce 3-substituted isoindolinones, has been investigated. A route starting with a nitroaldol (... |
|
|
Bifunctional phase-transfer catalysis in the asymmetric synthesis of biologically active isoindolinones.
Beilstein J. Org. Chem. 11 , 2591-9, (2015) New bifunctional chiral ammonium salts were investigated in an asymmetric cascade synthesis of a key building block for a variety of biologically relevant isoindolinones. With this chiral compound in ... |
|
|
The aldol addition of readily enolizable 1, 3-dicarbonyl compounds to 2-cyanobenzaldehyde in the synthesis of novel 3-substituted isoindolinones. More V, et al.
Synthesis 2011(18) , 3027-31, (2011)
|
| o-Cyanobenzaldehyde |
| 2-cyanobenzaldehyde |
| 2-Formylbenzonitrile |
| MFCD00017503 |
| Benzonitrile, 2-formyl- |
| 2-Formylbenzolcarbonitril |
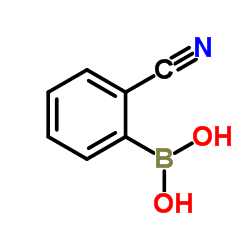 CAS#:138642-62-3
CAS#:138642-62-3 CAS#:201230-82-2
CAS#:201230-82-2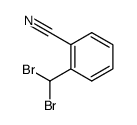 CAS#:655-63-0
CAS#:655-63-0 CAS#:22115-41-9
CAS#:22115-41-9 CAS#:2042-37-7
CAS#:2042-37-7 CAS#:643-79-8
CAS#:643-79-8 CAS#:529-19-1
CAS#:529-19-1 CAS#:557-21-1
CAS#:557-21-1![(1Z)-1-[(4-methoxyphenyl)imino]-1H-isoindol-3-amine structure](https://image.chemsrc.com/caspic/076/104830-22-0.png) CAS#:104830-22-0
CAS#:104830-22-0 CAS#:3839-22-3
CAS#:3839-22-3 CAS#:52670-38-9
CAS#:52670-38-9 CAS#:4152-92-5
CAS#:4152-92-5 CAS#:4456-77-3
CAS#:4456-77-3 CAS#:6525-45-7
CAS#:6525-45-7 CAS#:6587-24-2
CAS#:6587-24-2 CAS#:3297-72-1
CAS#:3297-72-1 CAS#:36078-60-1
CAS#:36078-60-1 CAS#:5398-11-8
CAS#:5398-11-8
