Spiperone

Spiperone structure
|
Common Name | Spiperone | ||
|---|---|---|---|---|
| CAS Number | 749-02-0 | Molecular Weight | 395.47 | |
| Density | N/A | Boiling Point | 630.6ºC at 760 mmHg | |
| Molecular Formula | C23H26FN3O2 | Melting Point | 190-193.6ºC | |
| MSDS | Chinese USA | Flash Point | 335.2ºC | |
| Symbol |


GHS07, GHS08 |
Signal Word | Warning | |
Use of SpiperoneSpiperone is a potent dopamine D2, serotonin 5-HT1A, and serotonin 5-HT2A antagonist. Spiperone is a widely used pharmacological tool. Spiperone has the potential for the research of neurology diseases[1]. |
| Name | spiperone |
|---|---|
| Synonym | More Synonyms |
| Description | Spiperone is a potent dopamine D2, serotonin 5-HT1A, and serotonin 5-HT2A antagonist. Spiperone is a widely used pharmacological tool. Spiperone has the potential for the research of neurology diseases[1]. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 630.6ºC at 760 mmHg |
|---|---|
| Melting Point | 190-193.6ºC |
| Molecular Formula | C23H26FN3O2 |
| Molecular Weight | 395.47 |
| Flash Point | 335.2ºC |
| Exact Mass | 395.20100 |
| PSA | 52.65000 |
| LogP | 3.54880 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H361 |
| Precautionary Statements | P281 |
| Personal Protective Equipment | Eyeshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | Xn |
| Risk Phrases | 22-63 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | XX8925000 |
|
~91% 
Spiperone CAS#:749-02-0 |
| Literature: Zigelboim, Isaac; Weissberg, Avi; Cohen, Yoram Journal of Organic Chemistry, 2013 , vol. 78, # 14 p. 7001 - 7012 |
|
~45% 
Spiperone CAS#:749-02-0 |
| Literature: Hashizume, Kazunari; Hashimoto, Naoto; Miyake, Yoshihiro Bulletin of the Chemical Society of Japan, 1997 , vol. 70, # 3 p. 681 - 687 |
|
~% 
Spiperone CAS#:749-02-0 |
| Literature: Bulletin of the Chemical Society of Japan, , vol. 70, # 3 p. 681 - 687 |
|
~% 
Spiperone CAS#:749-02-0 |
| Literature: Bulletin of the Chemical Society of Japan, , vol. 70, # 3 p. 681 - 687 |
|
~% 
Spiperone CAS#:749-02-0 |
| Literature: Bulletin of the Chemical Society of Japan, , vol. 70, # 3 p. 681 - 687 |
|
~% 
Spiperone CAS#:749-02-0 |
| Literature: Bulletin of the Chemical Society of Japan, , vol. 70, # 3 p. 681 - 687 |
|
~% 
Spiperone CAS#:749-02-0 |
| Literature: Bulletin of the Chemical Society of Japan, , vol. 70, # 3 p. 681 - 687 |
| Precursor 7 | |
|---|---|
| DownStream 8 | |
|
The herbicide glyphosate causes behavioral changes and alterations in dopaminergic markers in male Sprague-Dawley rat.
Neurotoxicology 46 , 79-91, (2015) Glyphosate (Glyph) is the active ingredient of several herbicide formulations. Reports of Glyph exposure in humans and animal models suggest that it may be neurotoxic. To evaluate the effects of Glyph... |
|
|
Blonanserin extensively occupies rat dopamine D3 receptors at antipsychotic dose range.
J. Pharmacol. Sci. 127(3) , 326-31, (2015) Antagonism of the dopamine D3 receptor has been hypothesized to be beneficial for schizophrenia cognitive deficits, negative symptoms and extrapyramidal symptoms. However, recent animal and human stud... |
|
|
Fluoro-substituted phenylazocarboxamides: Dopaminergic behavior and N-arylating properties for irreversible binding.
Bioorg. Med. Chem. 23 , 3938-47, (2015) Phenylazocarboxamides can serve as bioisosteres for cinnamides, which are widely occurring substructures in medicinal chemistry. Starting from our lead compound 2, the introduction of additional fluor... |
|
Name: Evaluated for the binding affinity to hippocampus striatal membranes at 5-hydroxytryp...
Source: ChEMBL
Target: 5-hydroxytryptamine receptor 1A
External Id: CHEMBL615963
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Displacement of [3H]BTX-B from neuronal voltage-gated sodium channel in rat cerebral ...
Source: ChEMBL
Target: N/A
External Id: CHEMBL3101695
|
|
Name: Increase the activity of the Burkholderia fixLJ 2-component system
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: Burkholderia multivorans
External Id: HMS1625
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Antagonist activity at dopamine D3 receptor (unknown origin) expressed in CHOK1 cells...
Source: ChEMBL
Target: D(3) dopamine receptor
External Id: CHEMBL2390214
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
| 8-[4-(4-fluorophenyl)-4-oxobutyl]-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one |
| Spiroperidol |
| Espiperona |
| R 5147 |
| Spiperonum |
| Spiropitan |
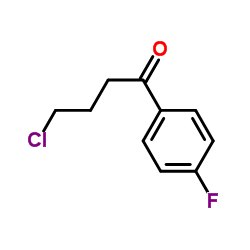
![1-Phenyl-1,3,8-triaza-spiro[4.5]decan-4-one structure](https://image.chemsrc.com/caspic/259/1021-25-6.png)

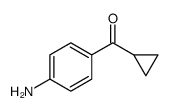
![N-[4-(4-chlorobutanoyl)phenyl]acetamide structure](https://image.chemsrc.com/caspic/311/56924-11-9.png)


![8-[4-(4-fluorophenyl)-4-oxobutyl]-1-phenyl-4-(2-fluoroethoxy)-1,3,8-triazaspiro[4.5]dec-3-ene structure](https://image.chemsrc.com/caspic/280/106114-43-6.png) CAS#:106114-43-6
CAS#:106114-43-6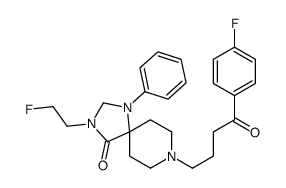 CAS#:106114-42-5
CAS#:106114-42-5![8-[4-(4-fluorophenyl)-4-oxobutyl]-1-phenyl-4-(3-fluoropropoxy)-1,3,8-triazaspiro[4.5]dec-3-ene structure](https://image.chemsrc.com/caspic/204/106114-45-8.png) CAS#:106114-45-8
CAS#:106114-45-8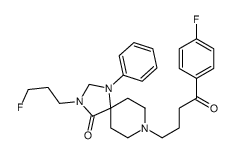 CAS#:106114-44-7
CAS#:106114-44-7![8-[4-(4-fluorophenyl)-4-oxobutyl]-1-phenyl-3-prop-2-enyl-1,3,8-triazaspiro[4.5]decan-4-one structure](https://image.chemsrc.com/caspic/332/102395-40-4.png) CAS#:102395-40-4
CAS#:102395-40-4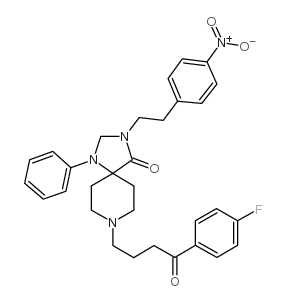 CAS#:136247-18-2
CAS#:136247-18-2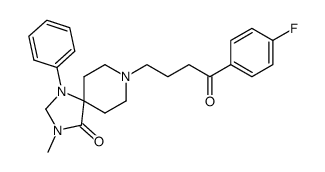 CAS#:87539-19-3
CAS#:87539-19-3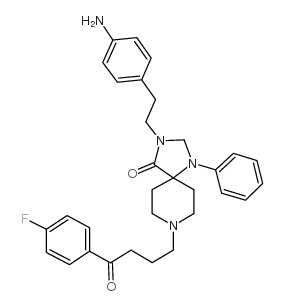 CAS#:93801-18-4
CAS#:93801-18-4