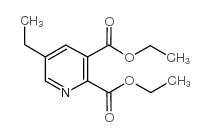
Imazethapyr
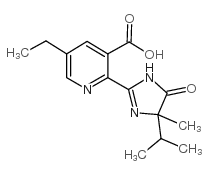
Imazethapyr structure
|
Common Name | Imazethapyr | ||
|---|---|---|---|---|
| CAS Number | 81335-77-5 | Molecular Weight | 289.33000 | |
| Density | 1.28 g/cm3 | Boiling Point | 446.8ºC at 760 mmHg | |
| Molecular Formula | C15H19N3O3 | Melting Point | 169-173ºC | |
| MSDS | Chinese USA | Flash Point | 224ºC | |
Use of ImazethapyrImazethapyr is an imidazolinone herbicide used in crops. Imazethapyr can protect crops from damage by weeds and annual grasses[1][2]. |
| Name | imazethapyr |
|---|---|
| Synonym | More Synonyms |
| Description | Imazethapyr is an imidazolinone herbicide used in crops. Imazethapyr can protect crops from damage by weeds and annual grasses[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Imazethapyr belongs to the imidazolinones family of herbicides that are being extensively used in a wide range of cropping systems to enhance crop yields and protect crops from damage by weeds and annual grasses in soybean and peanut. Imazethapyr would affect the transcription of photosynthesis-related genes and inhibit the antioxidant system of the plants and affect the chlorophyll synthesis[2]. |
| References |
| Density | 1.28 g/cm3 |
|---|---|
| Boiling Point | 446.8ºC at 760 mmHg |
| Melting Point | 169-173ºC |
| Molecular Formula | C15H19N3O3 |
| Molecular Weight | 289.33000 |
| Flash Point | 224ºC |
| Exact Mass | 289.14300 |
| PSA | 91.65000 |
| LogP | 1.39770 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| RTECS | US5682900 |
|
~86% 
Imazethapyr CAS#:81335-77-5 |
| Literature: Vasiliev, Aleksey N.; Lopez, Antonio F.; Fernandez, Julio D.; Mocchi, Anibal J. Molecules, 2004 , vol. 9, # 7 p. 535 - 540 |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
|
Enantioselective phytotoxicity of the herbicide imazethapyr and its effect on rice physiology and gene transcription.
Environ. Sci. Technol. 45(16) , 7036-43, (2011) Imazethapyr (IM) is a chiral herbicide and a widely used racemic mixture. This report investigated the enantioselectivity between R- and S-IM in rice and explored its causative mechanism at the physio... |
|
|
Molecular mechanism of enantioselective inhibition of acetolactate synthase by imazethapyr enantiomers.
J. Agric. Food Chem. 58(7) , 4202-6, (2010) Chiral compounds usually behave enantioselectively in phyto-biochemical processes. Imidazolinones are a class of chiral herbicides that are widely used. They inhibit branched-chain amino acid biosynth... |
|
|
Enantioselective phytotoxicity of the herbicide imazethapyr on the response of the antioxidant system and starch metabolism in Arabidopsis thaliana.
PLoS ONE 6(5) , e19451, (2011) The enantiomers of a chiral compound possess different biological activities, and one of the enantiomers usually shows a higher level of toxicity. Therefore, the exploration of the causative mechanism... |
| rac-5-ethyl-2-[(4R)-4-methyl-5-oxo-4-(propan-2-yl)-4,5-dihydro-1H-imidazol-2-yl]pyridine-3-carboxylic acid |
| MFCD00274561 |
| 2-[4,5-dihydro-4-methyl-4-(1-methylethyl)-5-oxo-1H-imidazol-2-yl]-5-ethyl-3-pyridinecarboxylic acid |
| 5-ethyl-2-[(RS)-4-isopropyl-4-methyl-5-oxo-2-imidazolin-2-yl]nicotinic acid |
| 5-ethyl-2-(4-methyl-5-oxo-4-propan-2-yl-1H-imidazol-2-yl)pyridine-3-carboxylic acid |
| 5-Ethyl-2-(4-isopropyl-4-methyl-5-oxo-4,5-dihydro-1H-imidazol-2-yl)nicotinic acid |