BAY 60-6583
Modify Date: 2025-08-23 15:14:07
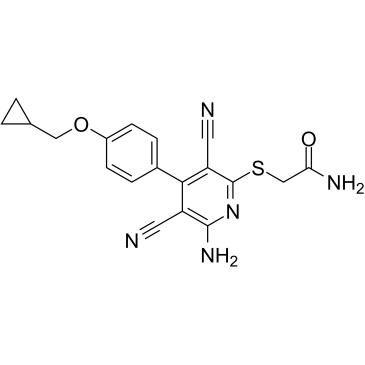
BAY 60-6583 structure
|
Common Name | BAY 60-6583 | ||
|---|---|---|---|---|
| CAS Number | 910487-58-0 | Molecular Weight | 379.43600 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C19H17N5O2S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of BAY 60-6583BAY 60-6583 is a potent and high-affinity agonist of adenosine A2B receptor (EC50 = 3 nM) over A1, A2A, and A3 receptors. BAY 60-6583 binds to mouse, rabbit, and dog A2BAR with Ki values of 750 nM, 340 nM and 330 nM, respectively. BAY 60-6583 has a cardioprotective effect in a myocardial ischemia model[1][5]. |
| Name | 2-[6-amino-3,5-dicyano-4-[4-(cyclopropylmethoxy)phenyl]pyridin-2-yl]sulfanylacetamide |
|---|---|
| Synonym | More Synonyms |
| Description | BAY 60-6583 is a potent and high-affinity agonist of adenosine A2B receptor (EC50 = 3 nM) over A1, A2A, and A3 receptors. BAY 60-6583 binds to mouse, rabbit, and dog A2BAR with Ki values of 750 nM, 340 nM and 330 nM, respectively. BAY 60-6583 has a cardioprotective effect in a myocardial ischemia model[1][5]. |
|---|---|
| Related Catalog | |
| In Vitro | BAY 60-6583 exhibits EC50 values for receptor activation >10,000 nM for both A1 and A2A AR and 3 nM for A2B AR subtype in CHO cells expressing recombinant human A1, A2A or A2B ARs[1]. BAY 60-6583(0-10 µM) exhibits the maximum agonist effect of BAY in the absence of siRNA is 68 %, which is significantly different from that in the presence of 5, 50 and 500 nM siRNA (54%, 48% and 36%, respectively). It exhibits EC50 values of BAY in the absence and presence siRNA with 98±22, 102±17, 127±31 and 93±19 nM, respectively, in T24 cells[3]. BAY 60-6583 (5 μM; 24 hours) increases the accumulation of cells at the G1 phase with a decrease in G2/M phase in RAW264.7 preosteoclasts[4]. BAY 60-6583 (5 μM; 24 hours) specifically inhibits the activation of Akt by M-CSF, whereas M-CSF-induced ERK1/2 activation is not affected by BAY 60-6583 treatment in RAW264.7 preosteoclasts[4]. Cell Cycle Analysis[4] Cell Line: RAW264.7 preosteoclasts Concentration: 5 μM Incubation Time: 48 hours Result: Caused an arrest of cells at the G1 phase. Western Blot Analysis[4] Cell Line: RAW264.7 preosteoclasts Concentration: 5 μM Incubation Time: 48 hours Result: Exhibited an inhibition of M-CSF-mediated Akt activation and resulted in the decrease of osteoclast proliferation. |
| In Vivo | BAY 60-6583 (intravenous injection; 100 mcg/kg) reduces the infarction area just prior to reperfusion in ischaemic rabbit hearts[1]. BAY 60-6583 (intraperitoneal injection; 2 mg/kg) attenuates LPS-induced lung injury, pre-treatment with this compound can significantly decrease LPS-increased IL-6 levels in WT-mice, In contrast, BAY 60-6583 treatment is ineffective in abrogating these inflammatory parameters in A2BAR−/− mice[2]. BAY 60-6583 (intratumoral administration) causes a significant increase in tumor-infiltrating MDSCs, it does not affect neither their ability to suppress T-cell proliferation nor their degree of maturation, it also stimulates the production of IL-10 and CCL2 in the tumor tissue[5]. Animal Model: A2BAR−/− mice on a C57BL/6J mice[1] Dosage: 2 mg/kg Administration: Intraperitoneal injection; 2 mg/kg Result: Demonstrated attenuation of lung inflammation and pulmonary edema in wild-type but not in gene-targeted mice for the A2BAR. |
| References |
| Molecular Formula | C19H17N5O2S |
|---|---|
| Molecular Weight | 379.43600 |
| Exact Mass | 379.11000 |
| PSA | 165.10000 |
| LogP | 4.17126 |
| InChIKey | ZTYHZMAZUWOXNC-UHFFFAOYSA-N |
| SMILES | N#Cc1c(N)nc(SCC(N)=O)c(C#N)c1-c1ccc(OCC2CC2)cc1 |
|
~98% 
BAY 60-6583 CAS#:910487-58-0 |
| Literature: BAYER HEALTHCARE AG Patent: WO2006/99958 A1, 2006 ; Location in patent: Page/Page column 15; 16 ; |
| Precursor 1 | |
|---|---|
| DownStream 0 | |
|
Name: This assay (PMID: 37558952) monitors the impact of a chemical compound on general aut...
Source: ChEMBL
Target: TERT-RPE1
External Id: CHEMBL5724699
|
|
Name: This assay (PMID: 37558952) monitors the impact of a chemical compound on general aut...
Source: ChEMBL
Target: U2OS
External Id: CHEMBL5724701
|
|
Name: Thermal Shift Assay. Domain: start/stop: M1-L298
Source: ChEMBL
Target: Cyclin-dependent kinase 2
External Id: CHEMBL5062802
|
|
Name: This assay (PMID: 37558952) monitors the impact of a chemical compound on ER-phagy fl...
Source: ChEMBL
Target: U2OS
External Id: CHEMBL5724700
|
|
Name: Thermal Shift Assay. Domain: start/stop: N44-E168
Source: ChEMBL
Target: Bromodomain-containing protein 4
External Id: CHEMBL5061006
|
|
Name: Human A3 receptor (Adenosine receptors)
Source: IUPHAR-DB
Target: A3 receptor (Adenosine receptors) [Homo sapiens]
External Id: 21_Human
|
|
Name: Thermal Shift Assay. Domain: start/stop: M26-R383
Source: ChEMBL
Target: Glycogen synthase kinase-3 beta
External Id: CHEMBL5065589
|
|
Name: Thermal Shift Assay. Domain: start/stop: S229-K512
Source: ChEMBL
Target: Tyrosine-protein kinase ABL1
External Id: CHEMBL5060015
|
|
Name: GPCR beta-arrestin recruitment assay with target: FPR2 Assay Type: PRESTO-Tango Conce...
Source: ChEMBL
Target: N-formyl peptide receptor 2
External Id: CHEMBL5209827
|
|
Name: GPCR beta-arrestin recruitment assay with target: FPR2 Assay Type: PRESTO-Tango Conce...
Source: ChEMBL
Target: N-formyl peptide receptor 2
External Id: CHEMBL5209826
|
Total 133, Current Page 1 of 14
1
2
3
4
5
| bay 60 c6583 |
| BAY 60-6583 |

