Pralidoxime Iodide
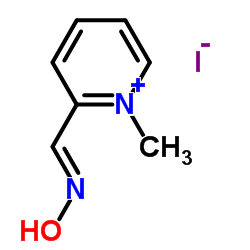
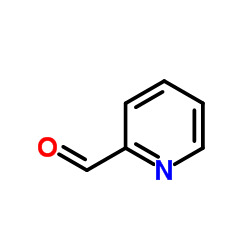
Pralidoxime Iodide structure
|
Common Name | Pralidoxime Iodide | ||
|---|---|---|---|---|
| CAS Number | 94-63-3 | Molecular Weight | 264.064 | |
| Density | 1.7439 g/ml | Boiling Point | N/A | |
| Molecular Formula | C7H9IN2O | Melting Point | 220 °C (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Pralidoxime IodidePralidoxime iodide is a reactivator of acetylcholinesterase (AChE). Pralidoxime iodide reactivates nerve agent, which inhibits AChE via direct nucleophilic attack by the oxime moiety on the phosphorus center of the bound nerve agent. Pralidoxime iodide is an antidote for organophosphate poisoning[1][2]. |
| Name | pralidoxime iodide |
|---|---|
| Synonym | More Synonyms |
| Description | Pralidoxime iodide is a reactivator of acetylcholinesterase (AChE). Pralidoxime iodide reactivates nerve agent, which inhibits AChE via direct nucleophilic attack by the oxime moiety on the phosphorus center of the bound nerve agent. Pralidoxime iodide is an antidote for organophosphate poisoning[1][2]. |
|---|---|
| Related Catalog | |
| In Vivo | Pralidoxime iodide (10-150 mg/kg; i.m.) reverses paraoxon-induced respiratory toxicity in mice[3]. Animal Model: Male F1B6D2 mice (toxic but non-lethal model of diethylparaoxon in awake, unrestrained mice)[3] Dosage: 10, 50, 100 and 150 mg/kg Administration: Intramuscularly Result: Reversal of diethylparaoxon-induced respiratory toxicity at 150 mg/kg. |
| References |
[2]. Eyer P, Buckley N. Pralidoxime for organophosphate poisoning. Lancet. 2006;368(9553):2110‐2111. |
| Density | 1.7439 g/ml |
|---|---|
| Melting Point | 220 °C (dec.)(lit.) |
| Molecular Formula | C7H9IN2O |
| Molecular Weight | 264.064 |
| Exact Mass | 263.975952 |
| PSA | 36.47000 |
| InChIKey | QNBVYCDYFJUNLO-UHFFFAOYSA-O |
| SMILES | C[n+]1ccccc1C=NO.I |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UU4375000 |
|
~% 
Pralidoxime Iodide CAS#:94-63-3 |
| Literature: Bioorganic and Medicinal Chemistry, , vol. 21, # 18 p. 5923 - 5930 |
|
~79% 
Pralidoxime Iodide CAS#:94-63-3 |
| Literature: Hampl, Frantisek; Mazac, Jiri; Liska, Frantisek; Spogl, Jiri; Kabrt, Lubomir; Suchanek, Miloslav Collection of Czechoslovak Chemical Communications, 1995 , vol. 60, # 5 p. 883 - 893 |
|
~% 
Pralidoxime Iodide CAS#:94-63-3 |
| Literature: Journal of the American Chemical Society, , vol. 79, p. 481,483 |
|
A trivalent approach for determining in vitro toxicology: Examination of oxime K027.
J. Appl. Toxicol. 35(2) , 219-27, (2014) Unforeseen toxic effects contribute to compound attrition during preclinical evaluation and clinical trials. Consequently, there is a need to correlate in vitro toxicity to in vivo and clinical outcom... |
|
|
Reactions of methylphosphonic difluoride with human acetylcholinesterase and oximes--Possible therapeutic implications.
Toxicol. Lett. 231(1) , 92-8, (2014) Highly toxic organophosphorus (OP) nerve agents are well characterized regarding chemical, biological and toxicological properties and the effectiveness of standard atropine and oxime therapy. Open li... |
|
|
An in vivo zebrafish screen identifies organophosphate antidotes with diverse mechanisms of action.
J. Biomol. Screen. 18(1) , 108-15, (2013) Organophosphates are a class of highly toxic chemicals that includes many pesticides and chemical weapons. Exposure to organophosphates, either through accidents or acts of terrorism, poses a signific... |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Flow Cytometric HTS Screen for inhibitors of the ABC transporter ABCB6 for Validation...
Source: NMMLSC
Target: ATP-binding cassette sub-family B member 6, mitochondrial [Homo sapiens]
External Id: UNMCMD_ABCB6_1o_ValidationSet
|
|
Name: Primary Screen Inhibitors of CD40 Signaling in BL2 Cells Measured in Cell-Based Syste...
Source: Broad Institute
Target: N/A
External Id: 7124-01_Inhibitor_SinglePoint_HTS_Activity
|
| 2-Pyridinealdoxime methiodide |
| 2-hydroxyiminomethyl-1-methylpyridinium iodide |
| protopamiodide |
| EINECS 202-349-6 |
| 1-methyl-2-hydroxyiminomethylpyridinium iodide |
| Pyridinium, 2-[(E)-(hydroxyimino)methyl]-1-methyl-, iodide (1:1) |
| pralidoximemethiodide |
| PRALIDOXIMELODIDE |
| PYRIDINE-2-CARBOXALDOXIME METHIODIDE |
| PRALIDOXIME IODIDE |
| MFCD00011982 |
| pyridin-2-aldoxin |
| p-2-am |
| 2-PAM |
| 2-pamiodide |
| 2-[(E)-(Hydroxyimino)methyl]-1-methylpyridinium iodide |
| 2-[(hydroxyimino)methyl]-1-methylpyridinium iodide |
 CAS#:14609-74-6
CAS#:14609-74-6